Enhanced electrochemical performance of copper-doped cobalt oxide nanowire-modified graphite felt as positive electrode material for vanadium redox flow batteries
銅摻雜氧化鈷奈米線改性石墨氈作為釩氧化還原流電池正極材料之電化學效能提升
📄 英文摘要
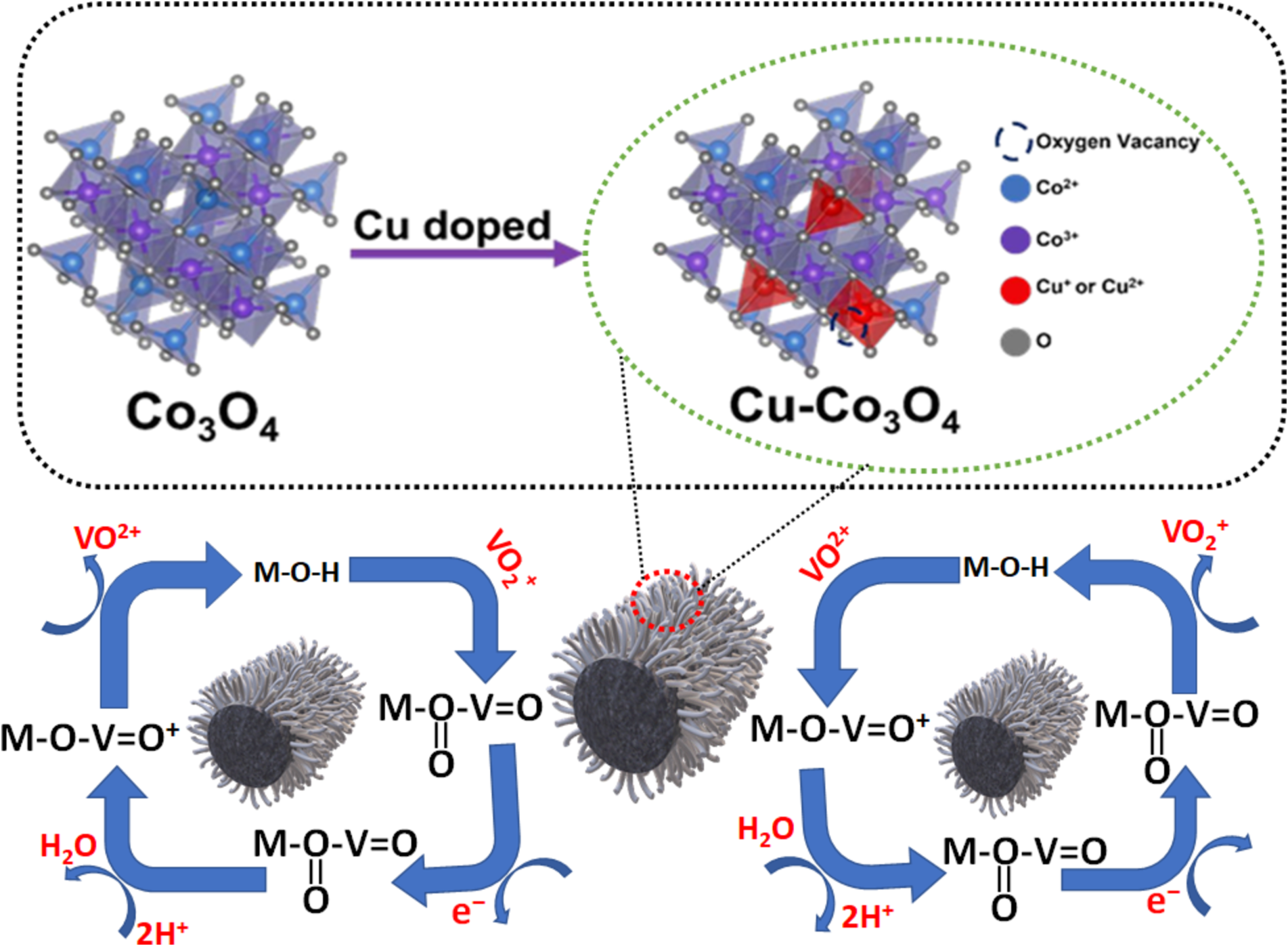
This research investigates copper (Cu) doped cobalt oxide (Co₃O₄) as a catalyst for vanadium redox flow batteries (VRFBs). Cu-doped Co₃O₄ is prepared on a heat-treated graphite felt (Cu-Co₃O₄-HGF) electrode through hydrothermal and calcination, enhancing the electrochemical performance of the VO₂⁺/VO₂⁺ redox couple. Electrochemical analysis confirms that the Cu-Co₃O₄-HGF electrode demonstrates superior catalytic activity for vanadium redox reaction, evidenced by a substantial reduction in charge transfer resistance compared to the undoped Co₃O₄-HGF. At a higher current density of 200 mA cm⁻², the VRFB using the Cu-Co₃O₄-HGF electrode achieves an energy efficiency (EE) of 76.04 %. This marks an improvement of 20.82 %, 12.67 %, and 4.93 % compared to the pristine graphite felt (PGF, 55.22 %), heat-treated graphite felt (HGF, 63.37 %), and Co₃O₄-HGF (71.11 %) electrode, respectively. Moreover, no significant efficiency decay is observed even after 500 cycles of VRFB operation, indicating the excellent stability of the Cu-Co₃O₄-HGF electrode during prolonged cycling. The superior performance of the Cu-Co₃O₄-HGF electrode is mainly due to the synergistic effects of Cu and Co, abundant oxygen vacancies, enhanced hydrophilicity, relatively high surface area, and increased Co₃⁺ concentration in the structure of Cu-Co₃O₄. These properties collectively enhance electron transfer kinetics and provide abundant active sites for redox reactions. This research contributes valuable insights to developing advanced electrode materials for next-generation energy storage technologies.
📄 中文摘要
本研究探討以銅(Cu)摻雜氧化鈷(Co₃O₄)作為釩氧化還原流電池(VRFB)的催化劑。Cu摻雜Co₃O₄透過水熱法與煅燒製備於熱處理石墨氈上(Cu-Co₃O₄-HGF),以提升VO₂⁺/VO₂⁺氧化還原對的電化學效能。電化學分析確認Cu-Co₃O₄-HGF电极對釩氧化還原反應具有優異的催化活性,其電荷轉移電阻相較於未摻雜之Co₃O₄-HGF顯著降低。在200 mA cm⁻²的較高電流密度下,使用Cu-Co₃O₄-HGF electrode的VRFB能源效率(EE)達到76.04%,相較於原始石墨氈(PGF, 55.22%)、熱處理石墨氈(HGF, 63.37%)及Co₃O₄-HGF(71.11%)分別提升20.82%、12.67%及4.93%。此外,即使在500次VRFB操作循環後亦無觀察到明顯的效率衰減,顯示Cu-Co₃O₄-HGF electrode在長時間循環中具有優異的穩定性。Cu-Co₃O₄-HGF electrode的優異效能主要源自Cu與Co的協同效應、豐富的氧空缺,提升的親水性、較高的比表面積,以及Cu-Co₃O₄結構中增加的Co₃⁺濃度。這些特性共同提升了電子傳輸動力學,並為氧化還原反應提供豐富的活性位點。本研究為開發下一代能源儲存技術的先進 electrode 材料提供了寶貴的見解。
🔬 五項核心重要發現
📊 關鍵圖表