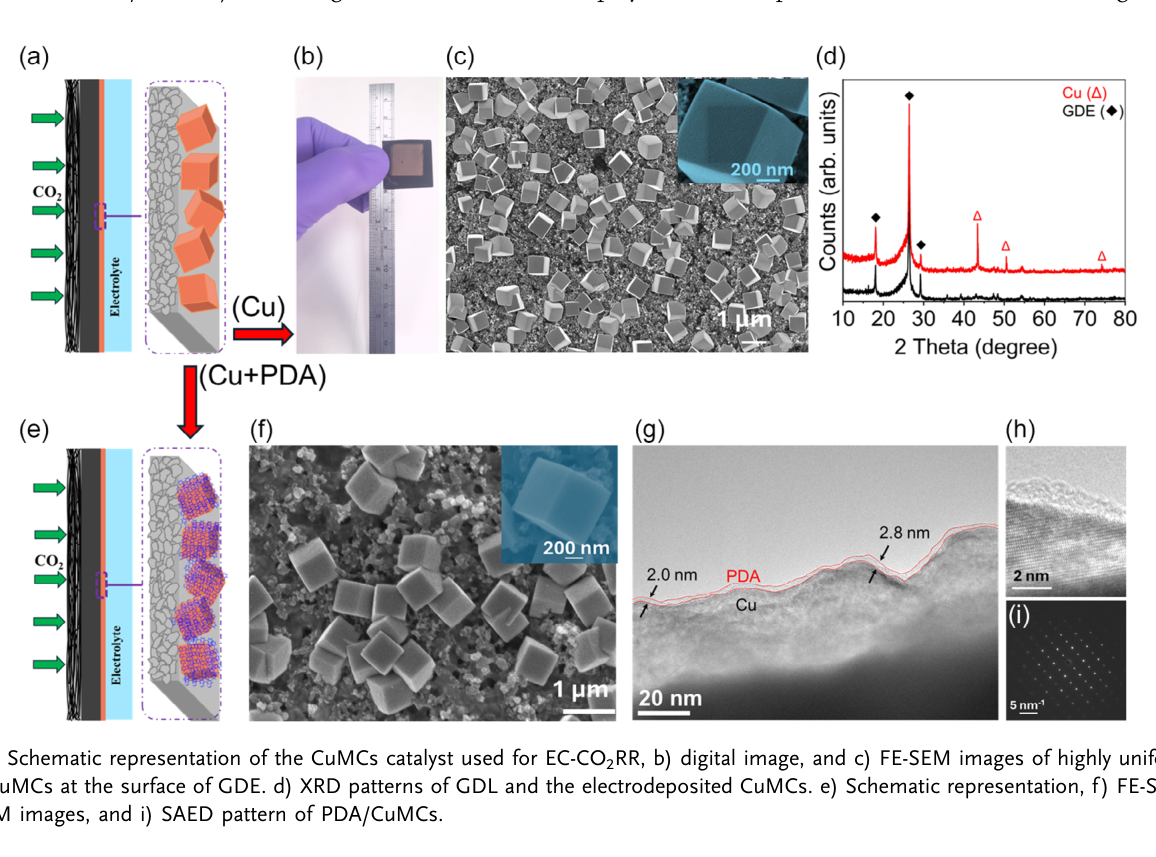
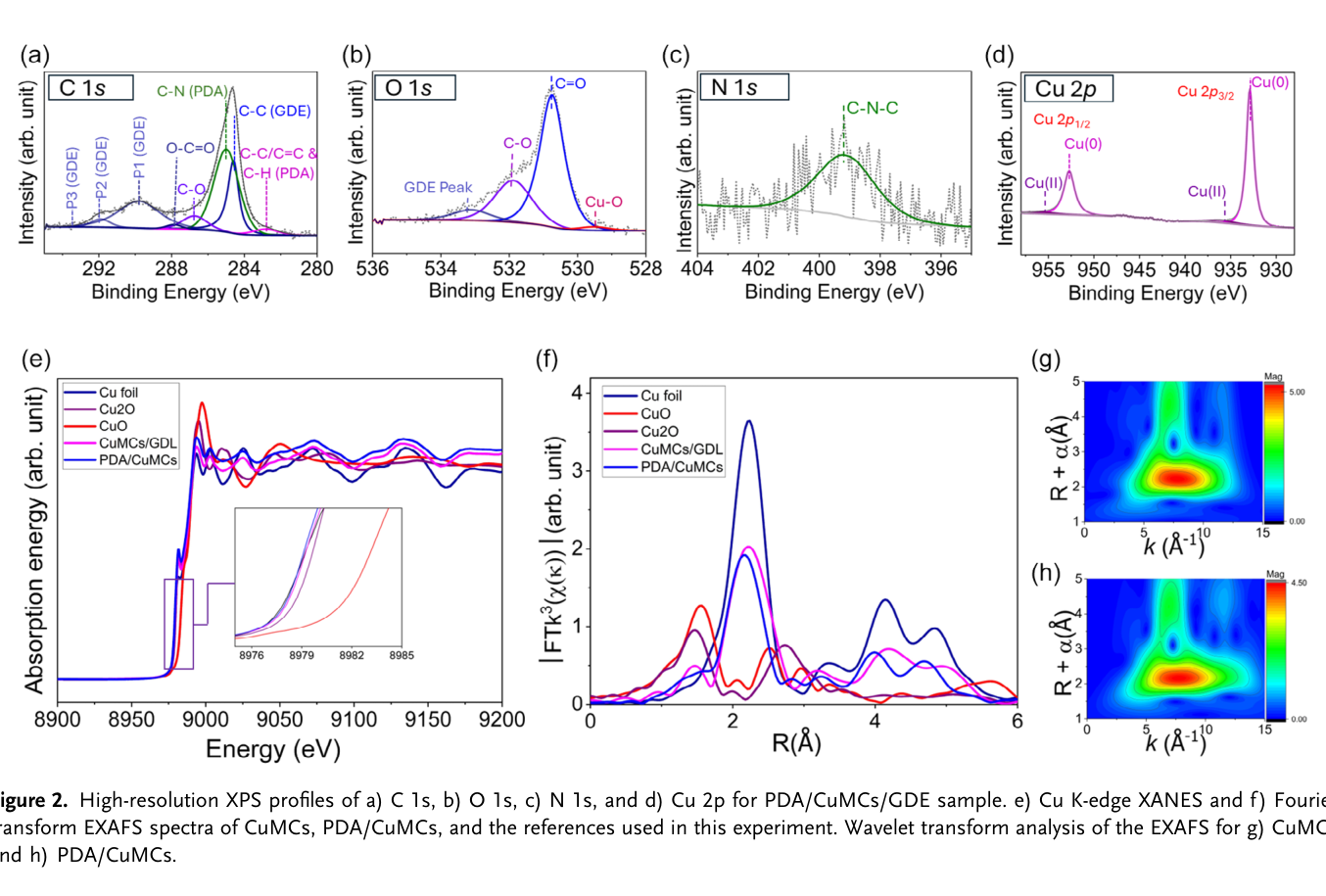
Restricting Copper Reconstruction with Ultrathin Polydopamine for Selective and Stable Electrochemical CO₂ Reduction Reaction to C₂ Products
📄 Abstract
A strategy is presented to mitigate copper surface reconstruction during electrochemical CO₂ reduction reaction (EC-CO₂RR) by conformally coating copper microcubes (CuMCs) with an ultrathin (2 nm) polydopamine (PDA) layer. The formation of nanocubes (30–50 nm in size) at the surface of microcubes at the early stages of the EC-CO₂RR contributes to suppressed surface reconstruction and sustained Faradaic efficiency (FE) for C₂ products. Furthermore, PDA coating effectively stabilizes adsorbed COatop and CObridge intermediates and promotes C─C coupling. As a result, the FE for C₂ products increases from 52.0 ± 4.0% for unmodified CuMCs to 81.6 ± 2.8% for PDA-coated CuMCs at −1.18 V versus reversible hydrogen electrode (VRHE). The PDA coating effectively slows down the surface evolution process of the catalyst during electrolysis. After 18 h of continuous operation at −0.88 VRHE, the CuMCs retain their original framework owing to a tightly adhered PDA layer that effectively stabilizes the surface and enhances catalyst durability. In situ surface-enhanced infrared absorption spectroscopy confirms the presence of adsorbed COatop, CObridge, and *OCCOH intermediates on PDA/CuMCs surface, which are believed to boost C─C coupling. This work highlights the potential of polymer film to stabilize the catalyst surface and steer product distribution in CO₂ electroreduction.
🔬 Five Key Findings
📊 Key Figures