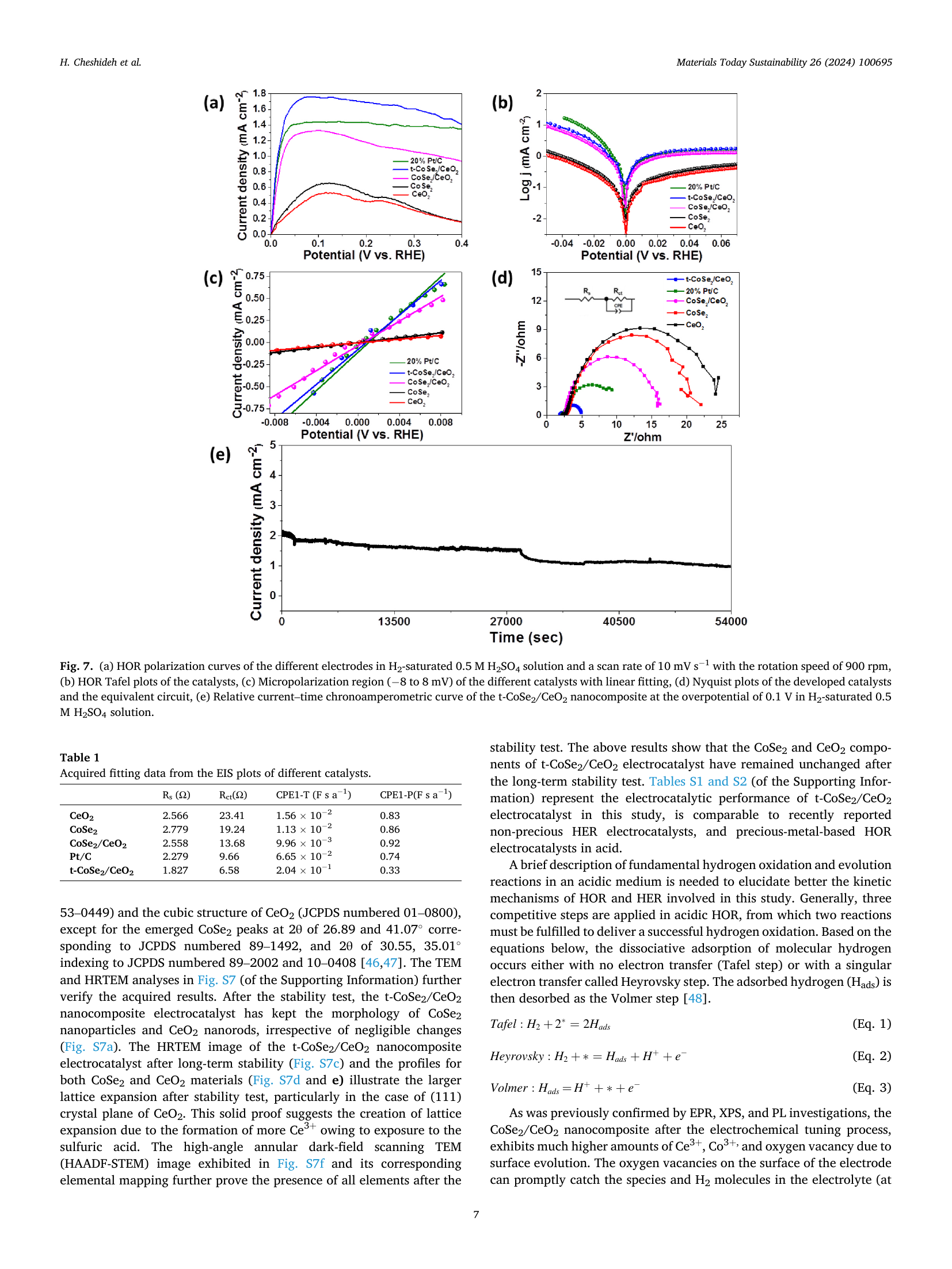
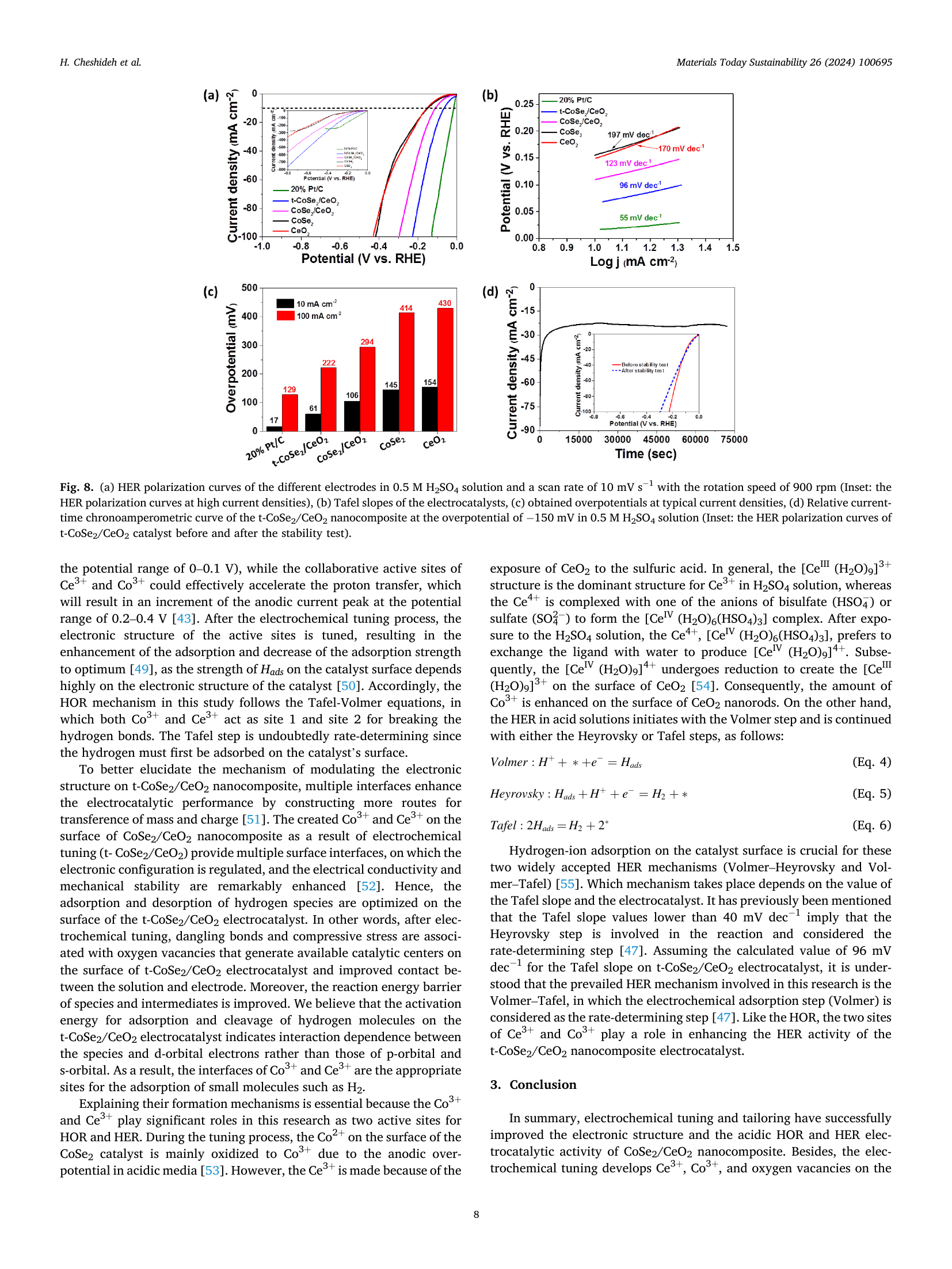
Electronic structure and defect density co-modulation of CoSe₂/CeO₂ nanocomposite for bifunctional hydrogen oxidation and reduction reactions
📄 Abstract
Developing bifunctional non-precious metal electrocatalysts toward hydrogen oxidation reaction (HOR) and hydrogen evolution reaction (HER) in acid is seriously challenged in various applications, such as hydrogen purifiers, fuel cells, and water electrolyzers. Here, we report the development of CoSe₂/CeO₂ nanocomposite electrocatalyst improved by electrochemical tuning for efficient HOR and HER in acid. Electrochemical tuning is shown to generate Ce³⁺, Co³⁺, and oxygen vacancies on the surface of the nanocomposite, which are considered as active sites for the adsorption and desorption of hydrogen species. The tuned catalyst (t-CoSe₂/CeO₂) indicates an impressive activity towards HOR and HER in 0.5 M H₂SO₄ solution, achieving an anodic current density of 1.8 mA cm⁻² at 900 rpm and a cathodic overpotential of 61 mV, respectively, outperforming the commercial Pt/C. Besides, the synthesized electrocatalyst delivers favorable HOR/HER durability. Our results suggest that the surface manipulation of CoSe₂/CeO₂ nanocomposite could provide promising insights for substituting precious electrocatalysts for HOR/HER.
🔬 Five Core Findings
📊 Key Figures