Electrodeposited barnacle-like phosphorized nickel–copper porous catalysts for oxygen evolution reaction
📄 Abstract
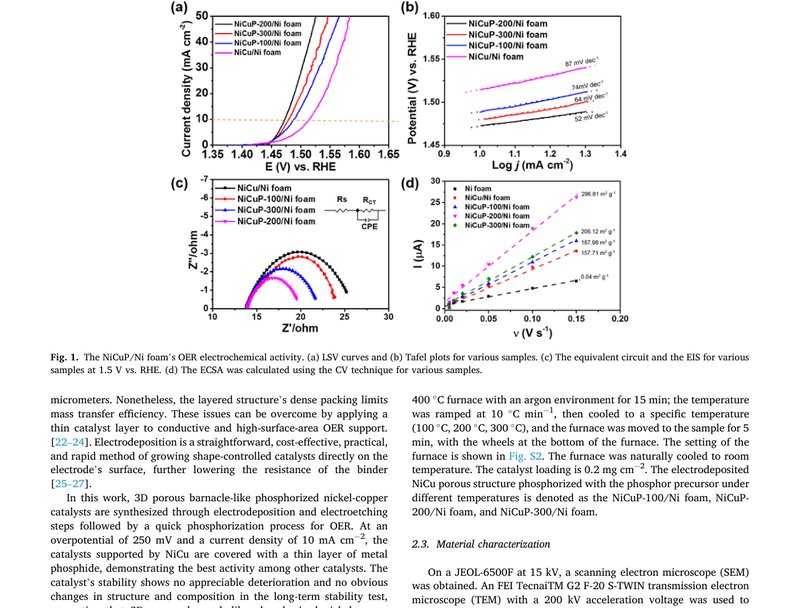
This work demonstrates the porous barnacle-like structure of NiCuP, which is co-electrodeposited on the nickel foam (NiCu/Ni foam) and does the phosphorization in a short time. Only the structure's surface can find phosphorus, which remains the conductivity of the nickel-copper and the porous structure (NiCuP-200/Ni foam). The catalysts show the overpotential of 250 mV for oxygen evolution reaction (OER) at the current density of 10 mA cm⁻². The cell using the NiCuP-200/Ni foam as the anode for the water electrolysis shows a highly consistent current density of about 180 mA cm⁻² at the cell potential of 1.8 V during 100 h operation. The phosphorization on the catalysts' surface helps the OER's activity. NiCuP-200/Ni foam remains porous after phosphorization, showing a high electrochemical surface area and low electrotransfer resistance.
🔬 Five Key Findings
📊 Key Figures