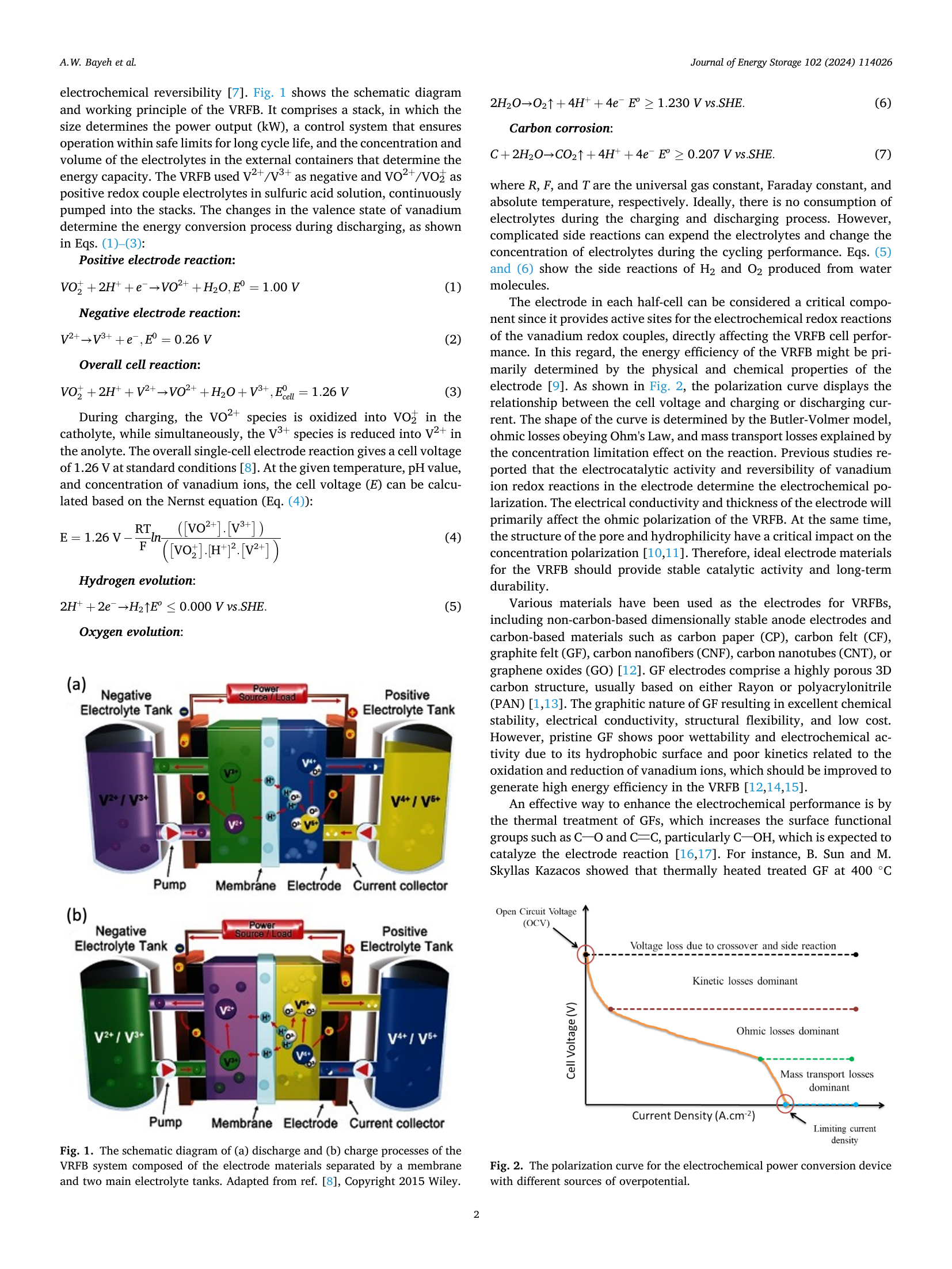
📄 Abstract
Vanadium redox flow batteries (VRFBs) are widely used in energy storage systems due to their large storage capacity and stable performance. As one of the critical components of VRFBs to provide the reaction sites for redox couples, an ideal electrode should possess excellent conductivity, electrochemical and chemical stability, good reaction kinetics, and a low price. Due to their favorable properties, carbon-based materials such as graphite felt (GF) and carbon paper (CP) are widely used as VRFB electrodes. However, these electrodes suffer from poor electrochemical activity towards VO2+/VO2+ and V2+/V3+ redox couples, caused by sluggish kinetics and high polarization, limiting the operation of VRFB at high current density. Specifically, the negative electrode is performance-limiting due to the V2+/V3+ reaction overlapping with the potential range of the hydrogen evolution reaction (HER), further hindering performance. Researchers have developed different strategies to improve the performance of VRFB electrodes towards the V2+/V3+ reaction. Here, the leading causes of capacity losses in VRFB towards the V2+/V3+ reaction, including the undesirable side reactions, such as the HER and degradation of carbon materials, are briefly reviewed. The electrochemical kinetics, the mechanism, and the role of various electrocatalysts in enhancing the V2+/V3+ redox reaction are discussed.
🔬 Five Core Findings
1
Activation of carbon felt (GF/CF) increases oxygen functional groups and active sites: Various techniques (especially thermal activation) are used to increase oxygen-containing functional groups on carbon fiber surfaces, which act as active sites for the vanadium redox process. However, high amorphous carbon structure, lower surface area, and insufficient oxygen functional groups favor hydrogen gas formation, which blocks active surface area and reduces V2+/V3+ electrochemical activity.
2
Carbon felt type selection is critical: PAN-based vs. Rayon-based GF: PAN-based felts show lower sp2:sp3 ratio than Rayon-based GF, resulting in decreased catalytic activity and conductivity, especially at the negative electrode. Rayon-based GF performs better at high current density, but the choice depends on performance requirements, cost, and scalability for commercial applications.
3
V2+/V3+ reaction competes with HER, causing electrolyte imbalance: The negative electrode potential is below the dynamic hydrogen reference electrode (NHE), allowing HER to occur simultaneously with V3+ reduction, leading to electrolyte imbalance and battery failure. Catalysts capable of inhibiting HER are needed to reduce side reactions and maintain cycling stability.
4
Metal oxide nanostructured electrocatalysts enhance negative electrode activity: Loading metal/metal oxide electrocatalysts on GF surface improves electrochemical activity toward V2+/V3+ redox couple. However, their performance is limited by low electronic conductivity, unstable combinations, long preparation routes, and inferior dispersion.
5
Future directions: defect engineering, doping, and metal oxide-carbon composites: Future research should develop advanced novel catalysts with superior catalytic activity, high conductivity, and low cost. Optimal dopant concentration creates moderate defect states and oxygen vacancies for V2+/V3+ active sites while inhibiting HER. Metal oxide-carbon composites combine advantages of both materials but need to overcome electrochemical incompatibility and capacity fade during cycling.