Nitrogen-doped carbonaceous electrode modified by biological metal-organic framework for vanadium redox flow batteries
📄 Abstract
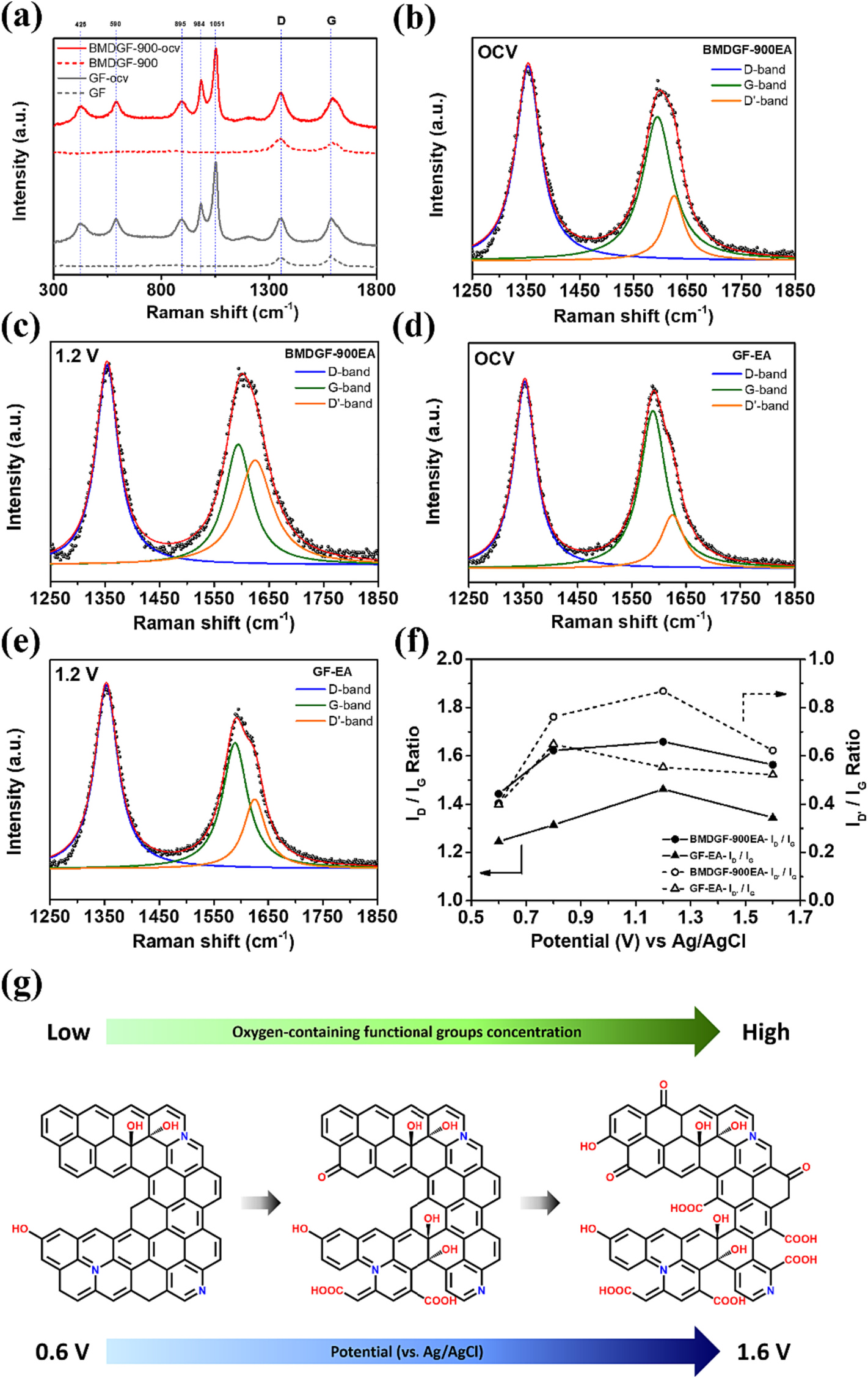
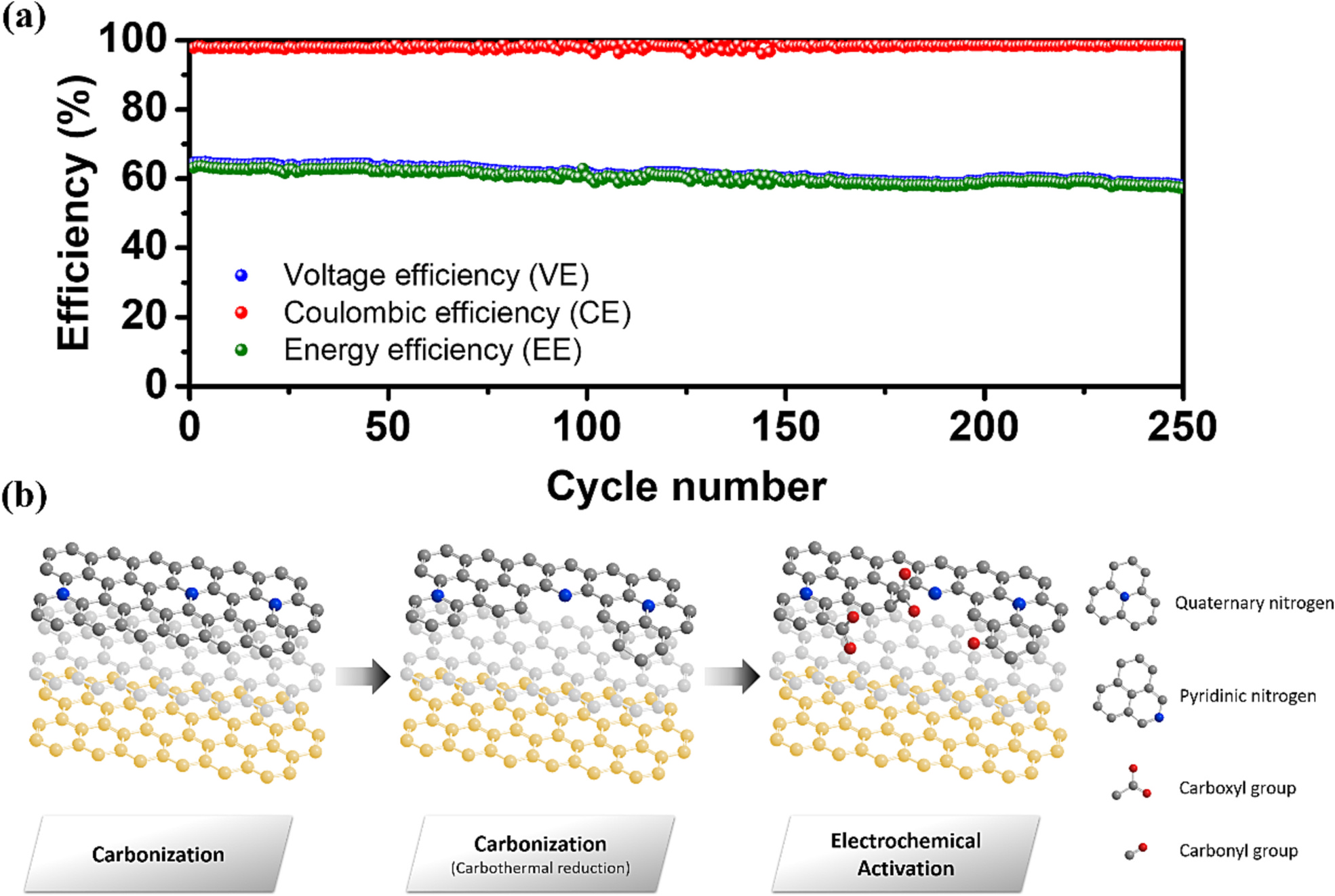
The biological metal-organic framework (Bio-MOF) is utilized to modify the carbonaceous electrode for the vanadium redox flow batteries. The microwave solvothermal method prepared the cobalt-based Bio-MOF on a graphite felt (GF) surface. Then, the nitrogen-doping graphite and porous structure formation were completed simultaneously during the carbonization process. Finally, electrochemical activation was conducted to remove cobalt metal from the GF electrode and raise the concentration of oxygen-containing functional group active sites. By in-situ Raman spectroscopy, the increase of disordered graphitic lattices could be observed by raising the potential from 0.6 V to 1.6 V. The microwave solvothermal method and electrochemical activation can effectively shorten the treatment time compared with the traditional solvothermal method and acidic etching processes. Among the carbonization parameters for Bio-MOF-covered GFs, maintaining the temperature at 900 °C for 2 h under an argon atmosphere is the most suitable. The annealed sample after electrochemical activation (BMDGF-900EA) displays 73.7 % energy efficiency at 120 mA cm⁻² and possesses a 90 % retention rate of efficiency after 250 cycles at 180 mA cm⁻². The superior performance results from more catalytic active sites, hydrophilic surface features, and larger specific surface area prove that this modification method derived from Bio-MOF indeed provides a novel and potential way to improve the VRFB performance.
🔬 Five Core Findings
📊 Key Figures