Enhanced electrochemical performance of copper-doped cobalt oxide nanowire-modified graphite felt as positive electrode material for vanadium redox flow batteries
📄 Abstract
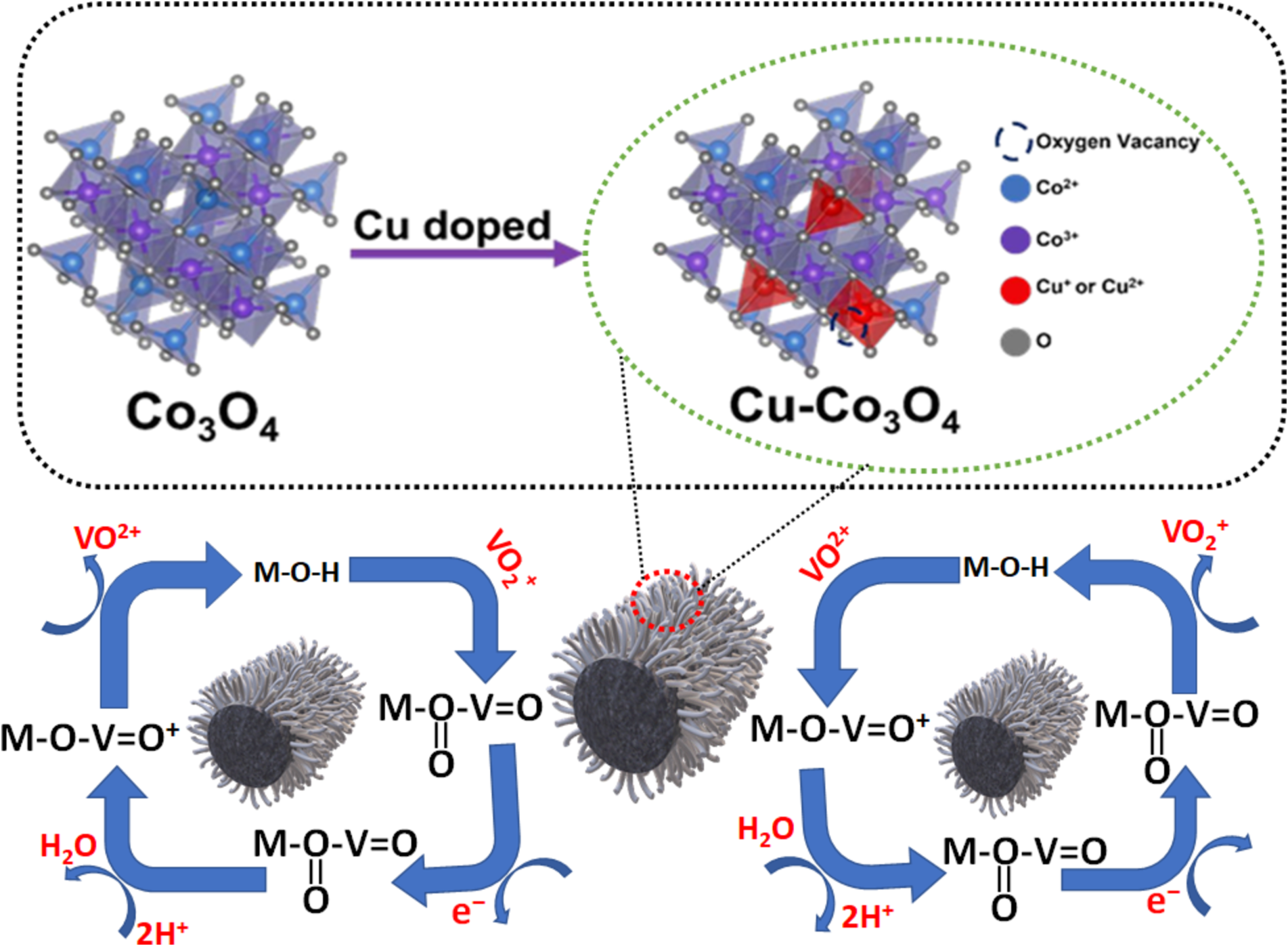
This research investigates copper (Cu) doped cobalt oxide (Co₃O₄) as a catalyst for vanadium redox flow batteries (VRFBs). Cu-doped Co₃O₄ is prepared on a heat-treated graphite felt (Cu-Co₃O₄-HGF) electrode through hydrothermal and calcination, enhancing the electrochemical performance of the VO₂⁺/VO₂⁺ redox couple. Electrochemical analysis confirms that the Cu-Co₃O₄-HGF electrode demonstrates superior catalytic activity for vanadium redox reaction, evidenced by a substantial reduction in charge transfer resistance compared to the undoped Co₃O₄-HGF. At a higher current density of 200 mA cm⁻², the VRFB using the Cu-Co₃O₄-HGF electrode achieves an energy efficiency (EE) of 76.04 %. This marks an improvement of 20.82 %, 12.67 %, and 4.93 % compared to the pristine graphite felt (PGF, 55.22 %), heat-treated graphite felt (HGF, 63.37 %), and Co₃O₄-HGF (71.11 %) electrode, respectively. Moreover, no significant efficiency decay is observed even after 500 cycles of VRFB operation, indicating the excellent stability of the Cu-Co₃O₄-HGF electrode during prolonged cycling. The superior performance of the Cu-Co₃O₄-HGF electrode is mainly due to the synergistic effects of Cu and Co, abundant oxygen vacancies, enhanced hydrophilicity, relatively high surface area, and increased Co₃⁺ concentration in the structure of Cu-Co₃O₄. These properties collectively enhance electron transfer kinetics and provide abundant active sites for redox reactions. This research contributes valuable insights to developing advanced electrode materials for next-generation energy storage technologies.
🔬 Five Core Findings
📊 Key Figures