Enhanced Electrochemical Performance of Vanadium Redox Flow Batteries Using Li₄Ti₅O₁₂/TiO₂ Nanocomposite-Modified Graphite Felt Electrodes
📄 Abstract
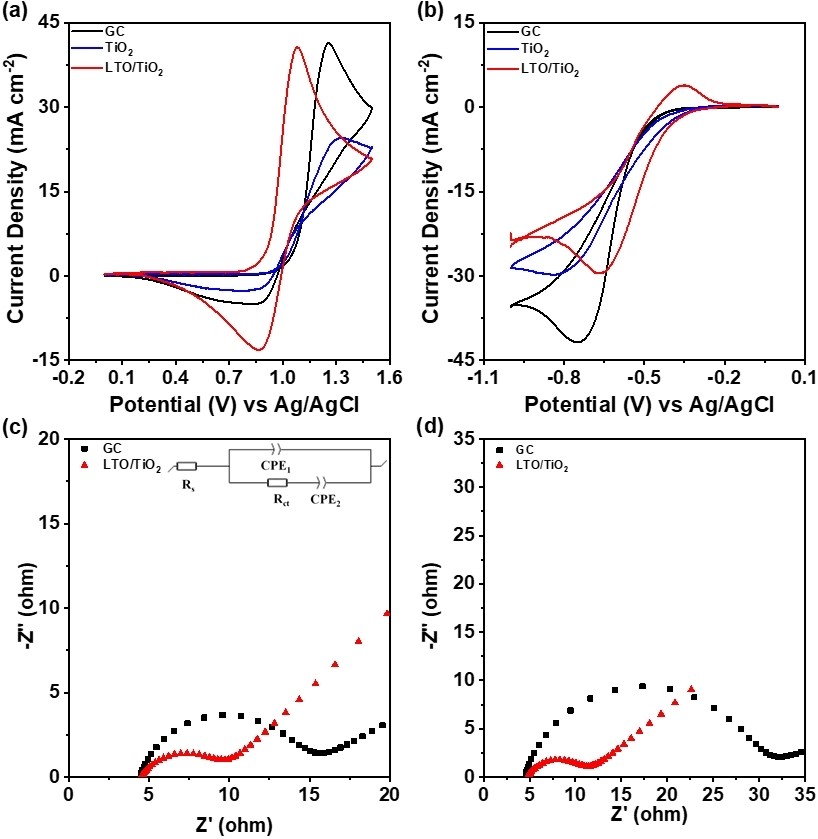
In this study, Li₄Ti₅O₁₂ (LTO) and TiO₂ nanocomposites uniformly were synthesized on the heat-treated graphite felt (HGF) through hydrothermal and heat treatment methods, denoted by LTO/TiO₂@HGF, which acts as effective electrocatalysts to enhance the electrochemical activity in vanadium redox flow battery (VRFB) systems. The cyclic voltammetry (CV) curves of the LTO/TiO₂@HGF show higher peak current densities and smaller peak separation than TiO₂@HGF, HGF, and pristine graphite felt (PGF) for catalyzing V²⁺/V³⁺ and VO₂⁺/VO²⁺, indicating superior electrochemical activity of LTO/TiO₂@HGF. The VRFB using LTO/TiO₂@HGF as the positive and negative electrodes demonstrates an energy efficiency of 82.89 % at 80 mA cm⁻². When the VRFB using LTO/TiO₂@HGF is applied at a high current density of 200 mA cm⁻², it still shows an energy efficiency of 62.22 %. However, the VRFB using PGF cannot perform any performance, and the VRFB using HGF only performs 51.94 %. This improvement can be attributed to the uniform distribution of LTO/TiO₂ nanowires on the surface of the graphite felt and the presence of oxygen vacancies on LTO/TiO₂, which increased the number of active sites for vanadium ion absorption.
🔬 Five Core Findings
📊 Key Figures