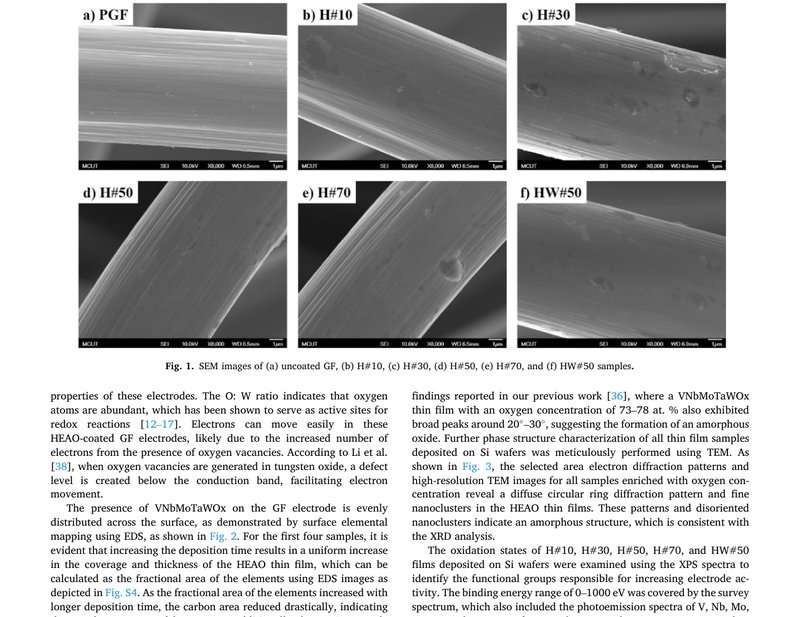
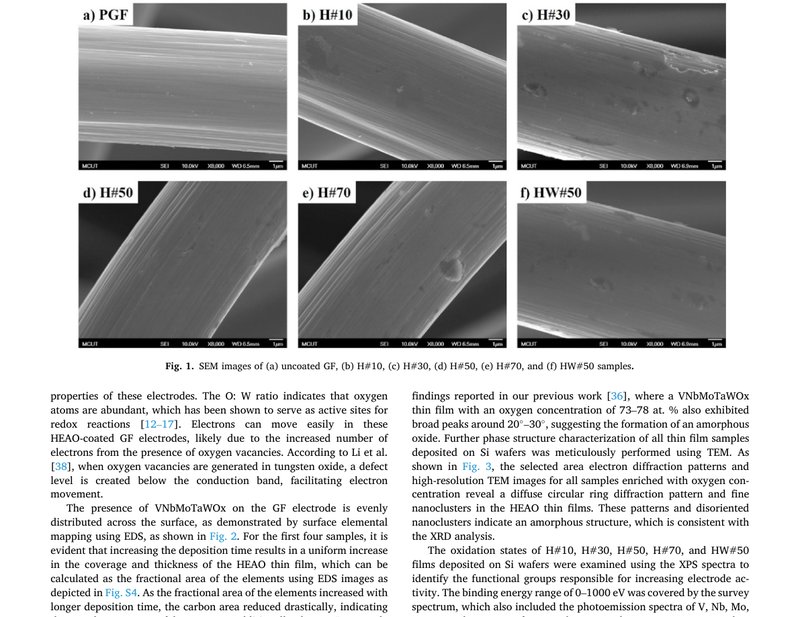
Unveiling the potential of oxygen-rich VNbMoTaWOx high-entropy catalyst for superior anode performance for sustainable vanadium redox flow batteries
📄 Abstract
High entropy alloy oxides (HEAOs) are a novel class of materials stabilized through configurational entropy, and they are anticipated to exhibit remarkable electrochemical properties. Here, we report the preparation of oxygen-rich amorphous VNbMoTaWOx HEAO thin films deposited on the surface of graphite felt (GF) electrode using a novel HiPIMS deposition technique intended for application in vanadium redox flow batteries (VRFBs). The modified GF electrode, featuring a 154 nm thick HEAO coating on the GF surface with an O/W ratio of 8.35, designated H#50, demonstrated superior electrocatalytic performance towards the VO⁺₂/VO₂⁺ redox couple, achieving energy efficiency values of 81.40 % at 100 mA cm⁻² and 71.40 % at 160 mA cm⁻². This represents a 7.70 % and 17.30 % improvement, respectively, over the uncoated GF electrode. The uniformly dispersed refractory metal, V, Nb, Mo, Ta, and W cations in the thin film, along with the optimal thickness, enable faster electron and ion transportation without obstructing the pores of GF. The diverse metal ions contribute to multiple oxidation states and abundant oxygen vacancies, which enhance the electroactive sites for the VO⁺₂/VO₂⁺ redox reaction. Additionally, this approach demonstrates remarkable cycling stability over 200 cycles.
🔬 Five Key Findings
📊 Key Figures