Engineering high-entropy oxide on reduced graphene oxide as a highly stable and efficient electrocatalyst for vanadium redox flow batteries
📄 Abstract
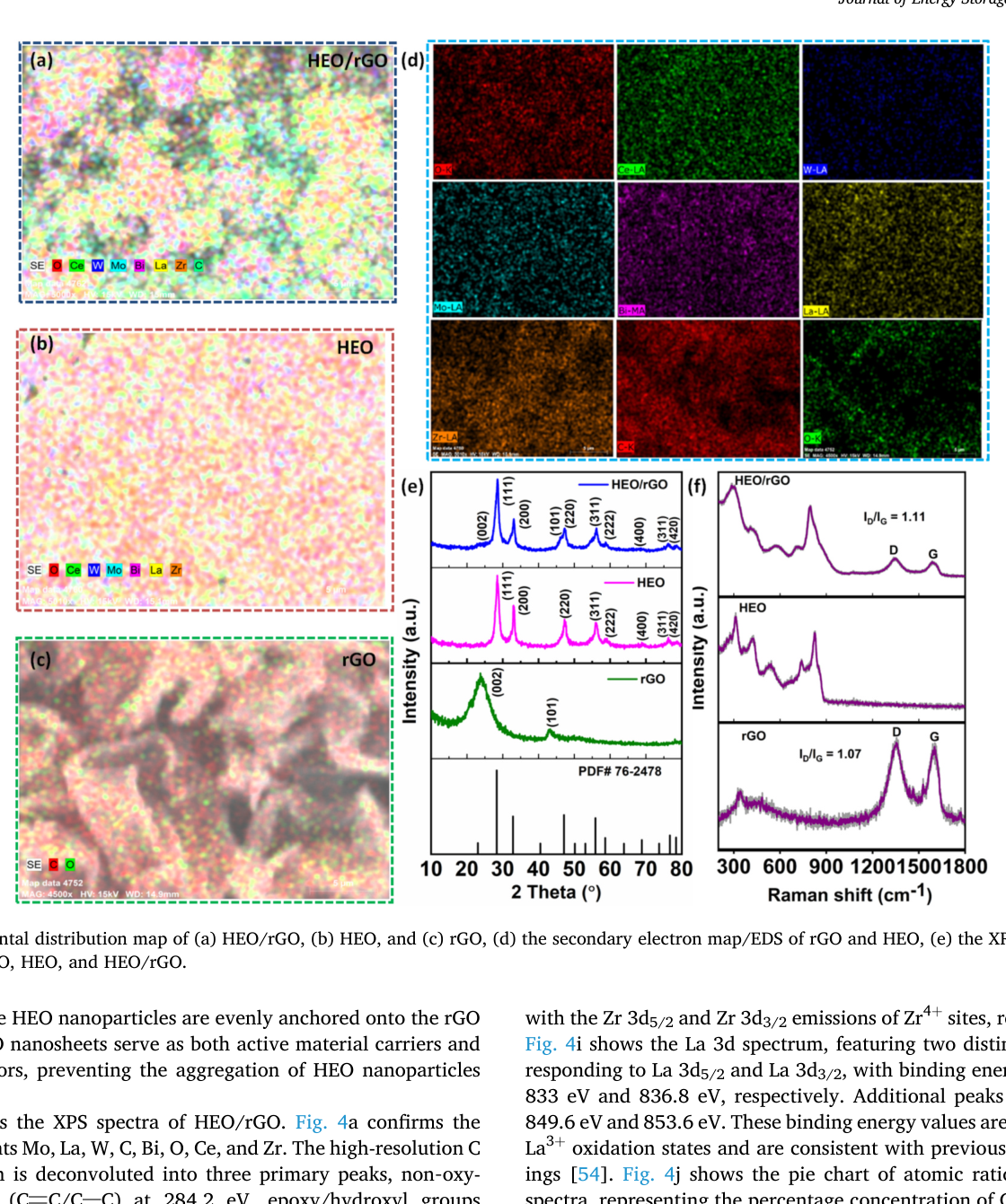
Efficient catalysts are essential for vanadium redox flow batteries (VRFBs), a key technology for large-scale energy storage. We report a high-entropy oxide of (CeMoBiWZrLa)O₂ supported by reduced graphene oxide ((CeMoBiWZrLa)O₂/rGO) as a composite catalyst for enhanced VRFB performance. The composite, synthesized via a hydrothermal reduction process, consists of dispersed HEO nanoparticles anchored on rGO sheets, which enhances electrolyte wettability and improves performance by enabling faster kinetics and more efficient ion transport. Moreover, the high configurational entropy, abundant oxygen vacancies, and inherent mechanical stability of HEO synergistically facilitate ion desorption, ensuring long-term durability. In-situ Raman spectroscopy reveals M–OH and C–OH₂⁺ intermediates, confirming accelerated redox kinetics in acidic electrolytes. The HEO/rGO-modified HGF electrode achieves energy efficiencies of 85.75% and 80.22% at 80 and 120 mA cm⁻² (improvements of 7.44% and 13.36%, respectively), with excellent stability over 300 cycles.
🔬 Five Key Findings
📊 Key Figures