Engineering palladium-nickel alloy sites on N-doped reduced graphene oxide for enhanced catalytic hydrogenation of vanadium electrolytes
📄 Abstract
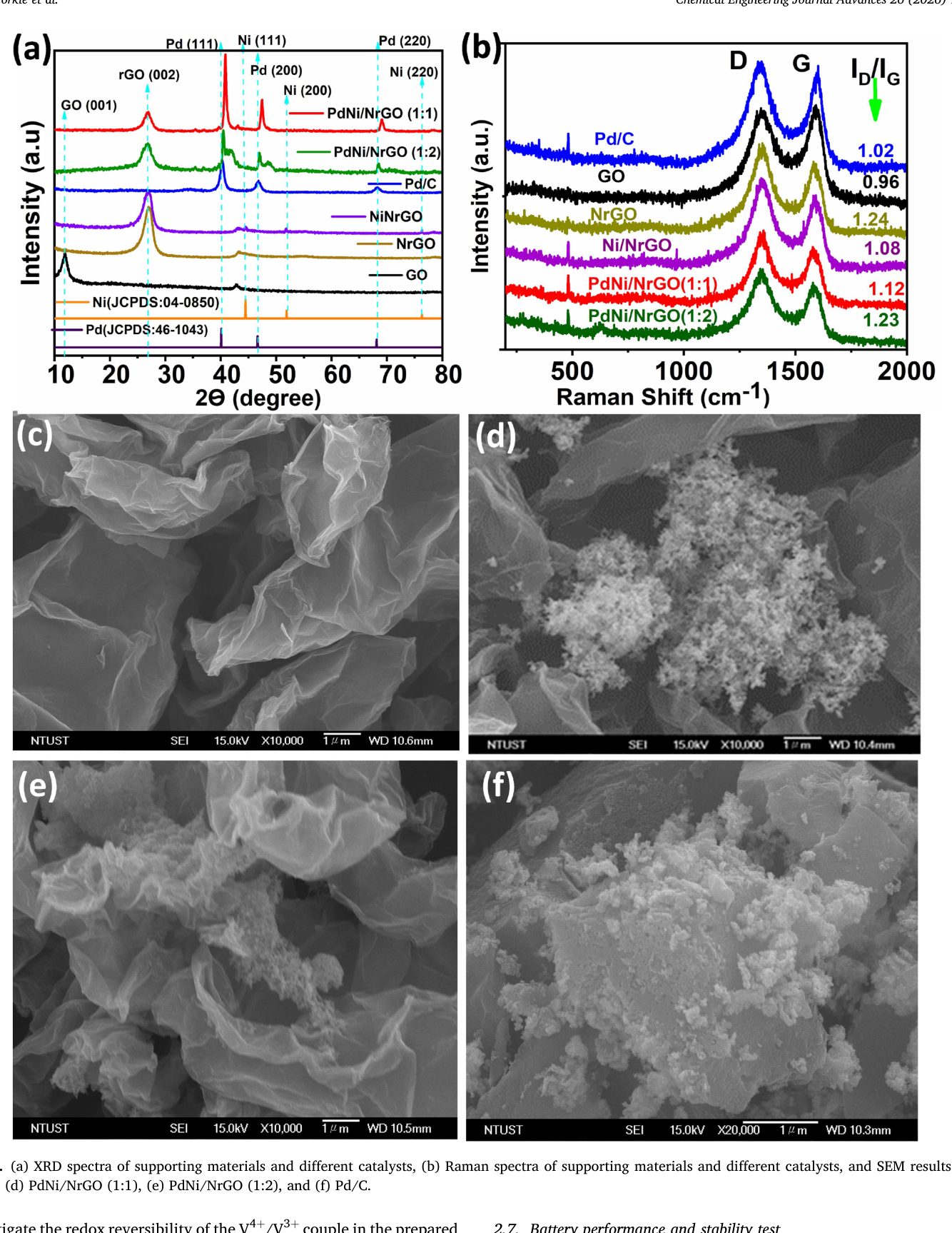
High-performance, low-cost electrolyte production remains a significant challenge for the commercialization of all-vanadium redox flow batteries (VRFBs). Herein, we demonstrate a robust strategy to engineer palladium-nickel (PdNi) alloy active sites on nitrogen-doped reduced graphene oxide (NrGO) for the efficient catalytic hydrogenation of VO₂⁺ (V⁴⁺) to the V³·⁵⁺. The resulting PdNi/NrGO catalyst outperforms commercial Pd/C, driven by synergistic Pd-Ni electronic interactions and the structural advantages of the N-doped support. Kinetic analysis reveals a remarkable turnover frequency (TOF) of 0.765 s⁻¹. The synthesized V³·⁵⁺ electrolyte delivers superior VRFB performance, achieving average efficiency gains of 16.6% (CE), 11.0% (EE), and 11.0% (VE) relative to the commercial baseline, while maintaining stable operation over 100 charge-discharge cycles. Furthermore, this bimetallic approach reduces catalyst costs by approximately 45% relative to commercial Pd/C and up to 71% compared to high-loading Pt-based systems.
🔬 Five Key Findings
📊 Key Figures