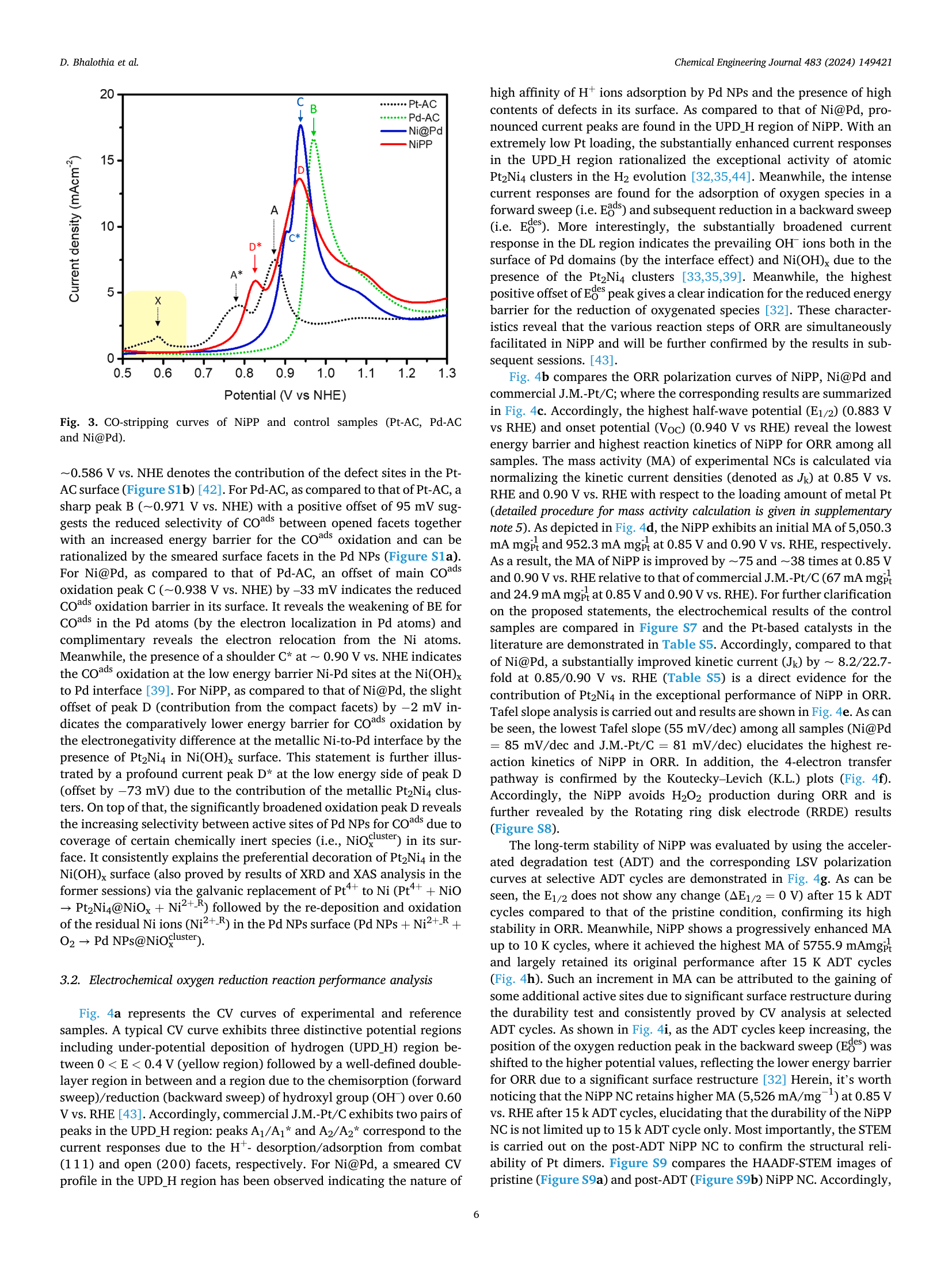
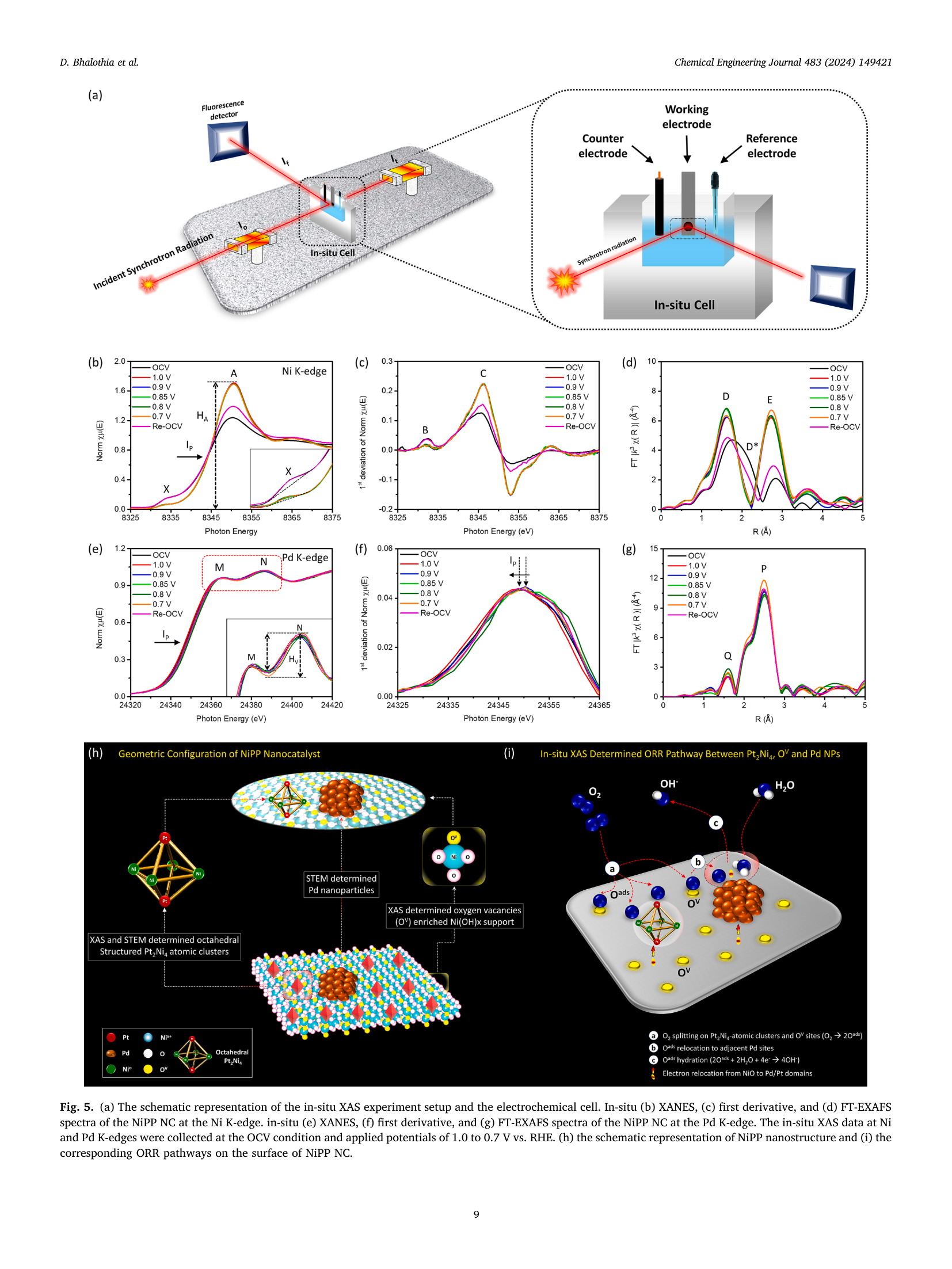
Potential synergy between Pt₂Ni₄ Atomic-Clusters, oxygen vacancies and adjacent Pd nanoparticles outperforms commercial Pt nanocatalyst in alkaline fuel cells
📄 Abstract
The oxygen reduction reaction (ORR) is a key "showstopper" for the commercial viability of alkaline fuel cells (AFCs). In this study, we developed a ternary nanocatalyst (NC) coloaded with Pt₂Ni₄ atomic clusters and palladium (Pd) nanoparticles (NPs) on oxygen vacancies (OVs) enriched nickel hydroxide-support (henceforth denoted as NiPP). This material outperforms the commercial J.M.-Pt/C (20 wt%) catalyst by ~75 and ~38 times with exceptionally high mass activities (MA)s of 5050.3 mAmg⁻¹Pt and 952.3 mAmg⁻¹Pt at 0.85 V and 0.90 V vs RHE, respectively, in alkaline ORR (0.1 M KOH). The high structural reliability of the Pt₂Ni₄ endows the NiPP NC with outstanding durability, where it achieves an optimum MA of 5755.9 mAmg⁻¹Pt after 10 K cycles in an accelerated durability test (ADT) and retains the original performance up to 15 K cycles. Moreover, NiPP delivers an outstanding power density (339.1 mW cm⁻²) and short circuit current density (910.8 mA cm⁻²), which exceeds the commercial J.M.-Pt/C (174.9 mW cm⁻² and 720.4 mA cm⁻²) when serving as the cathodic catalyst in an AFC. The results suggest that the exceptional performance originates from the potential synergism between the neighbouring reaction sites of Pt₂Ni₄ (OV in Ni(OH)x and Pd NPs). During ORR, the Pt₂Ni₄ atomic clusters and OVs boost the O₂ splitting, while the adjacent Pd NPs promote the subsequent relocation of OH⁻ ions.
🔬 Five Core Findings
📊 Key Figures