MOF-199-Based Nitrogen-Doped Bimetal Cathode Catalyst for Anion Exchange Membrane Fuel Cells
📄 Abstract
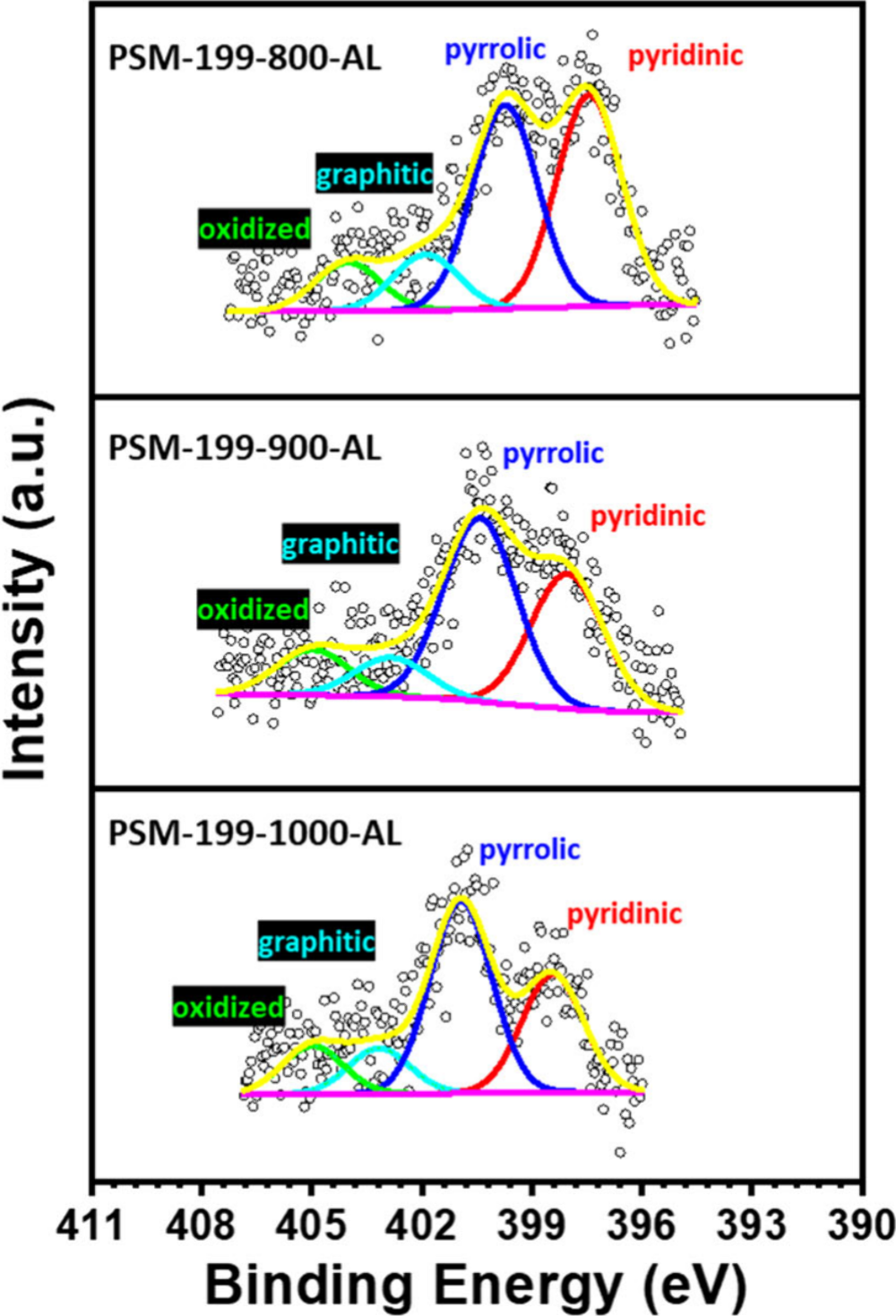
Anion exchange membrane fuel cells (AEMFCs) face significant challenges in developing cost-effective and high-performance cathode catalysts for the oxygen reduction reaction (ORR). This study reports a MOF-199-derived nitrogen-doped bimetal (Cu/Fe) carbon-based catalyst (PSM-199-900-AL) as a promising non-precious metal cathode catalyst for AEMFCs. The optimal thermal treatment at 900 °C yields a catalyst with exceptional ORR activity in alkaline media, achieving an onset potential of 0.99 V, a half-wave potential of 0.846 V, and a near-four-electron transfer pathway (n = 3.997). The Cu2+/Fe0 dual active sites work cooperatively: Fe activates O₂ while Cu facilitates intermediate desorption, synergistically enhancing ORR kinetics. When evaluated in an AEMFC, PSM-199-900-AL delivers a peak power density of 352.2 mW/cm², comparable to commercial Pt/C (381.9 mW/cm²). Moreover, the catalyst demonstrates outstanding durability with only a 30 mV half-wave potential loss after 30,000 cycles. This work highlights the great potential of MOF-derived bimetal catalysts as efficient and stable cathode materials for next-generation AEMFCs.
🔬 Five Core Findings
📊 Key Figures