Local collaboration between oxygen vacancy, palladium and platinum trimer triggers exceptional performance in the alkaline fuel cell
📄 Abstract
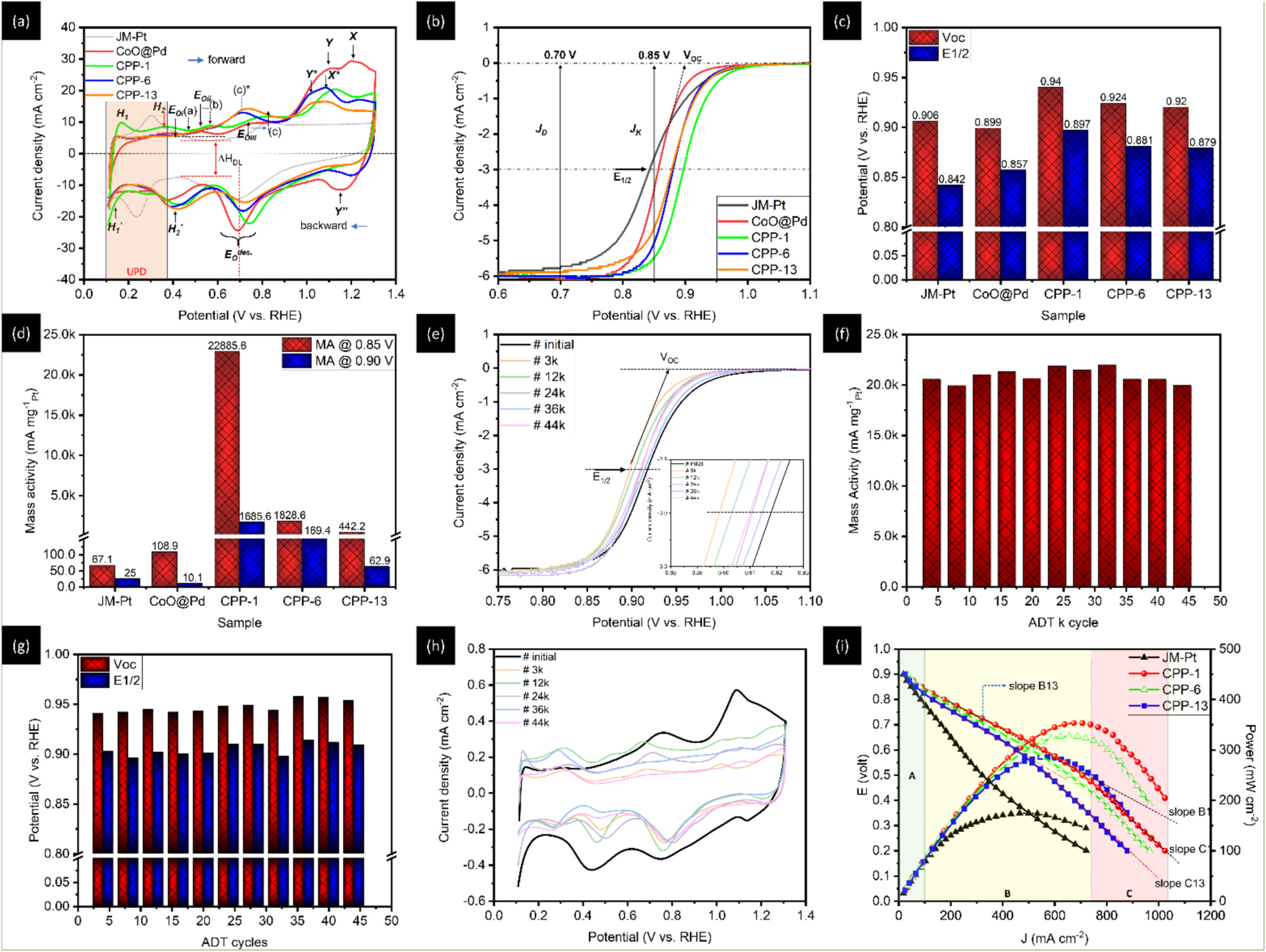
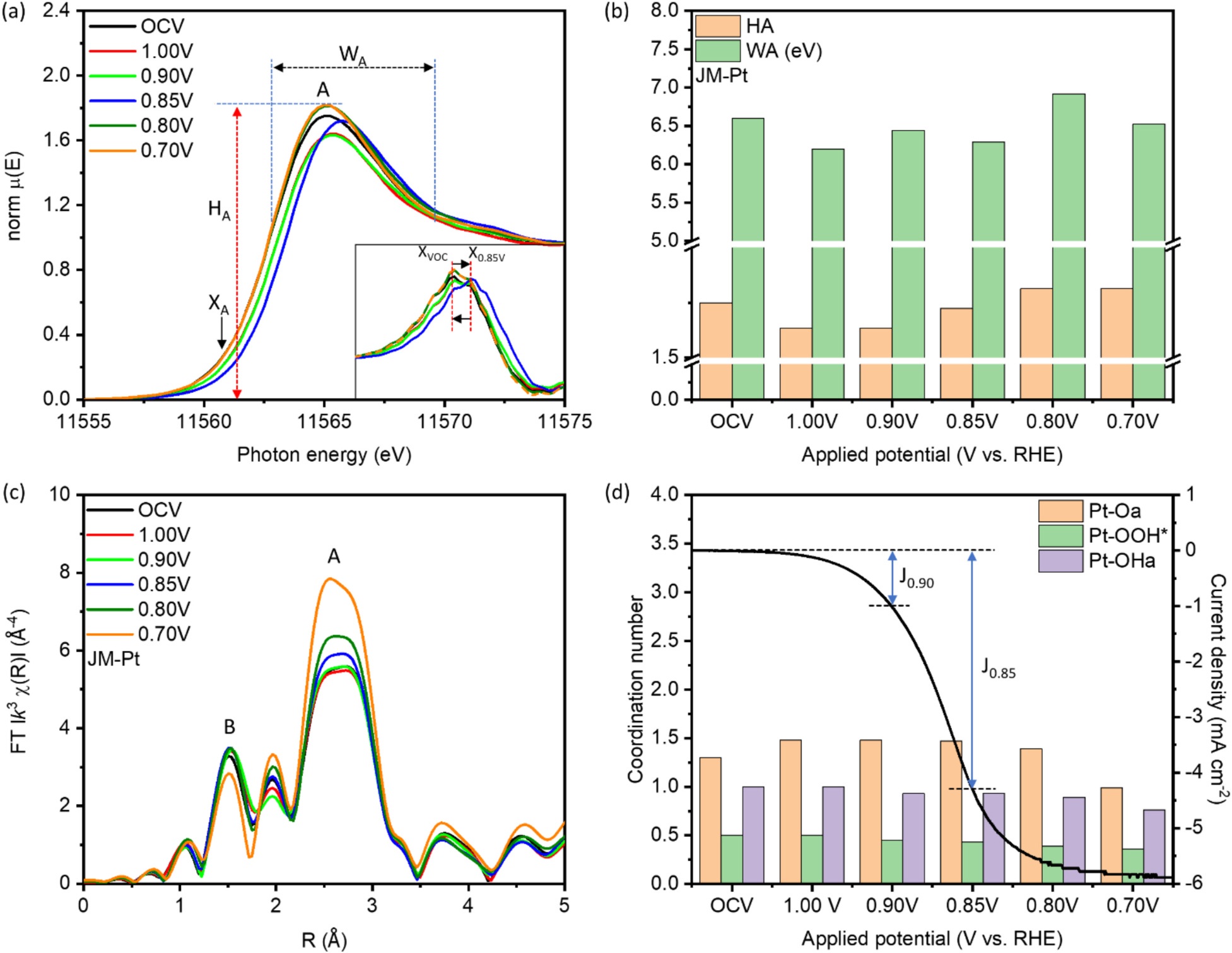
This study highlights a localized synergistic interaction among Pt trimers (PtT), oxygen vacancies (OV), and adjacent Pd atoms within a hierarchically structured catalyst. The catalyst, composed of cobalt oxide-supported palladium nanoparticles (NPs), is specifically optimized for the oxygen reduction reaction (ORR). With a remarkably low platinum loading of approximately 1 wt%, the catalyst (designated as CPP-1) demonstrates exceptional mass activities of 22,885.6 mA mgPt⁻¹ at 0.85 V and 1,685.6 mA mgPt⁻¹ at 0.90 V versus the reversible hydrogen electrode (RHE) in an alkaline (0.1 M KOH) ORR environment. These performance metrics surpass those of commercial platinum catalysts (J.M.-Pt/C; 20 wt% Pt) by factors of 341.6 and 71.7, respectively. Additionally, the PtT enable a maximum power output of 353.2 mW cm⁻² and a current density of 1,024.1 mA cm⁻² in an alkaline fuel cell (AFC) stack. In situ X-ray spectroscopy and electrochemical analysis reveal that PtT facilitates O₂ dissociation into chemisorbed oxygen (Oads), while Pd supports Oads diffusion, and oxygen vacancies (OV) drive the subsequent reduction steps. This coordinated mechanism ensures the simultaneous operation of all intermediate pathways and enables the regeneration of both active PtT sites and OV.
🔬 Five Core Findings
📊 Key Figures