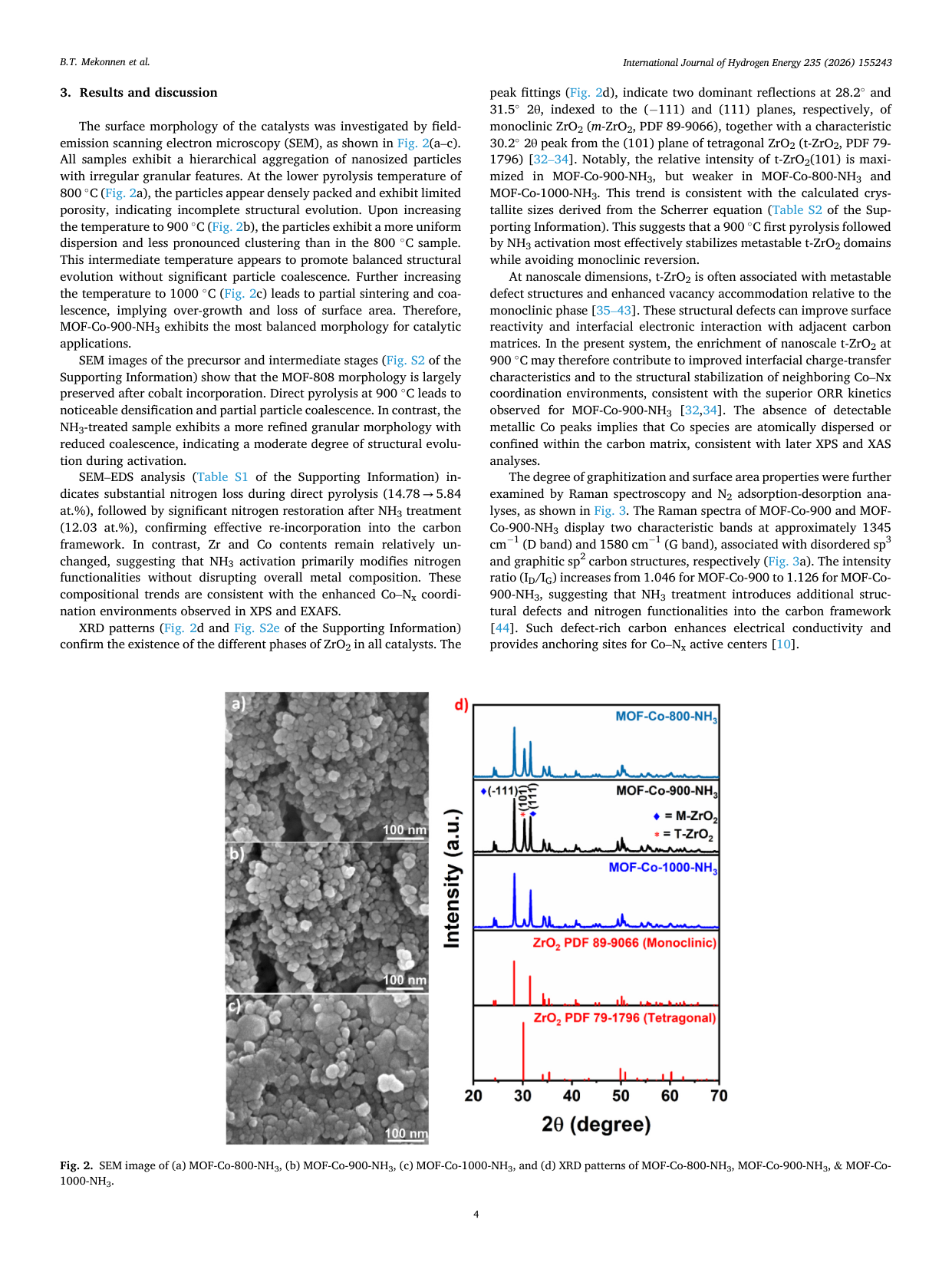
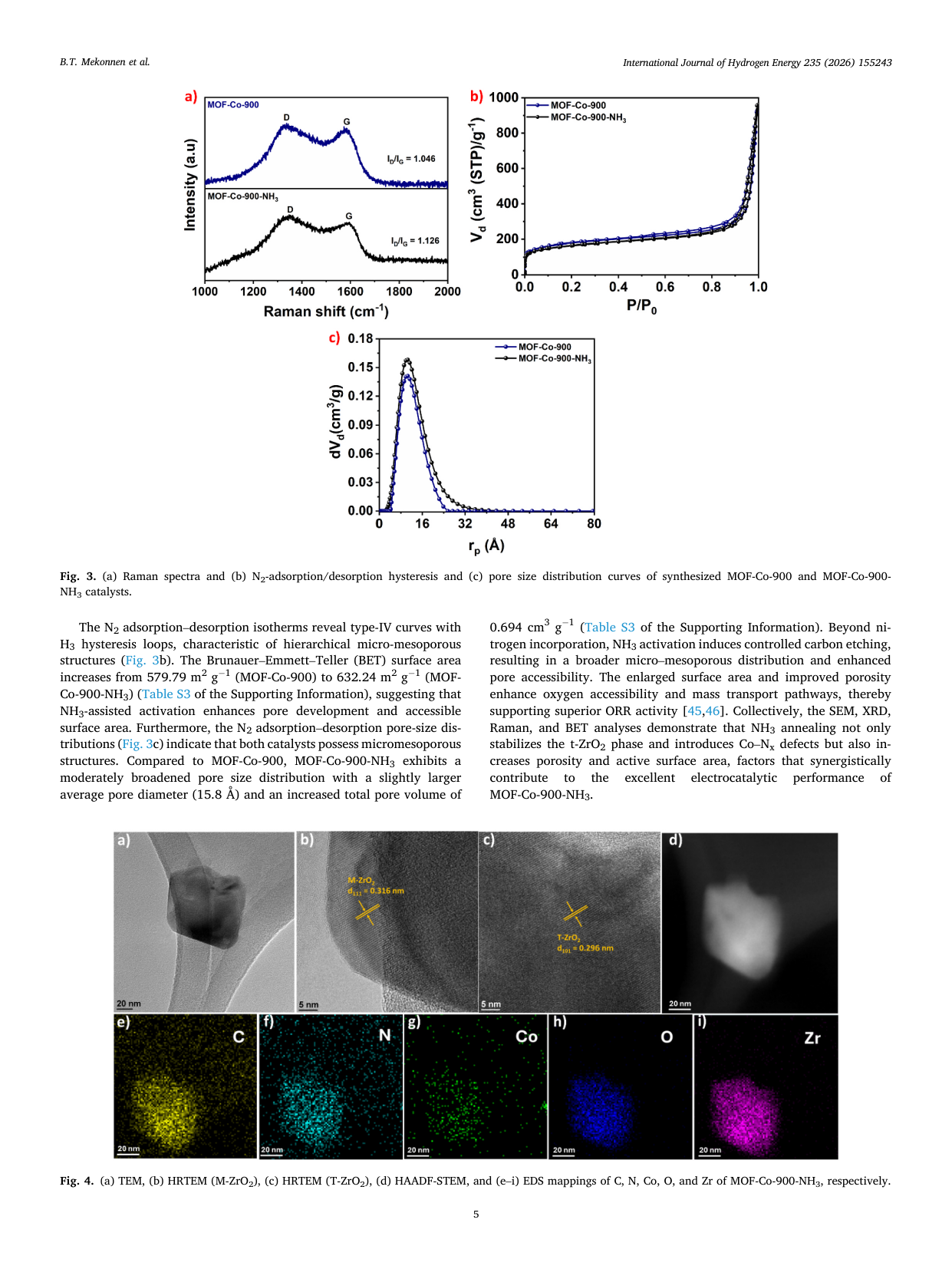
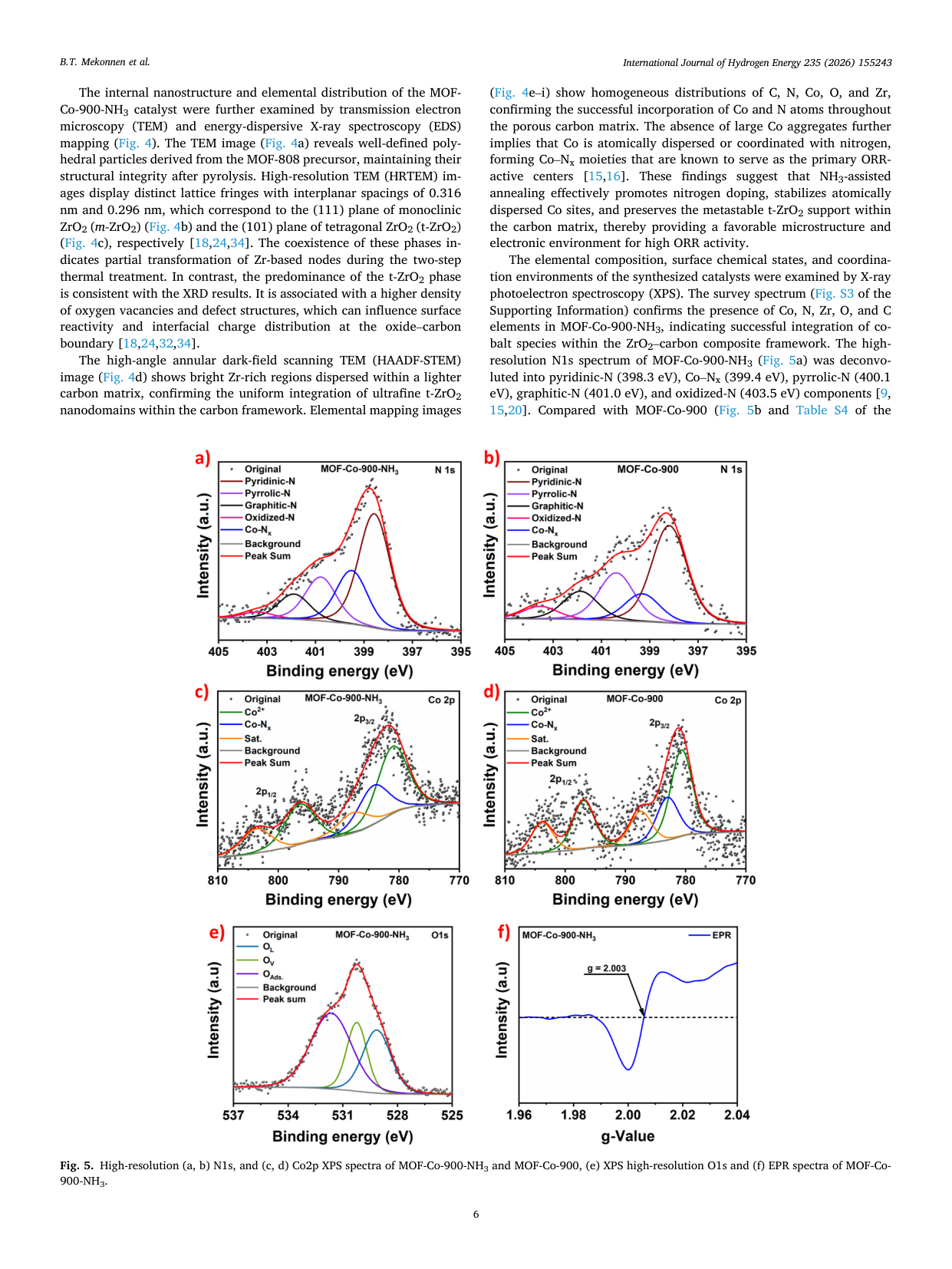
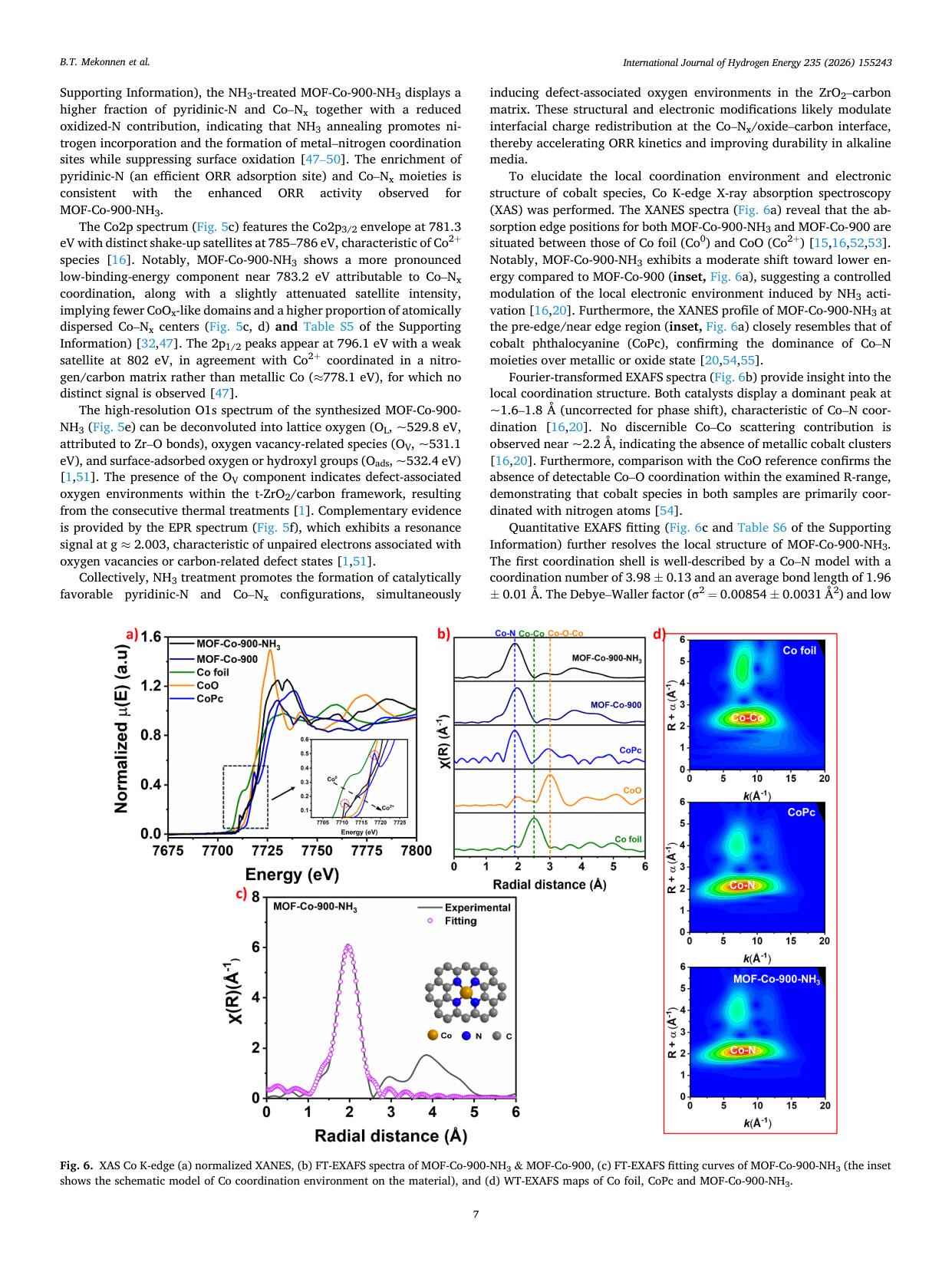
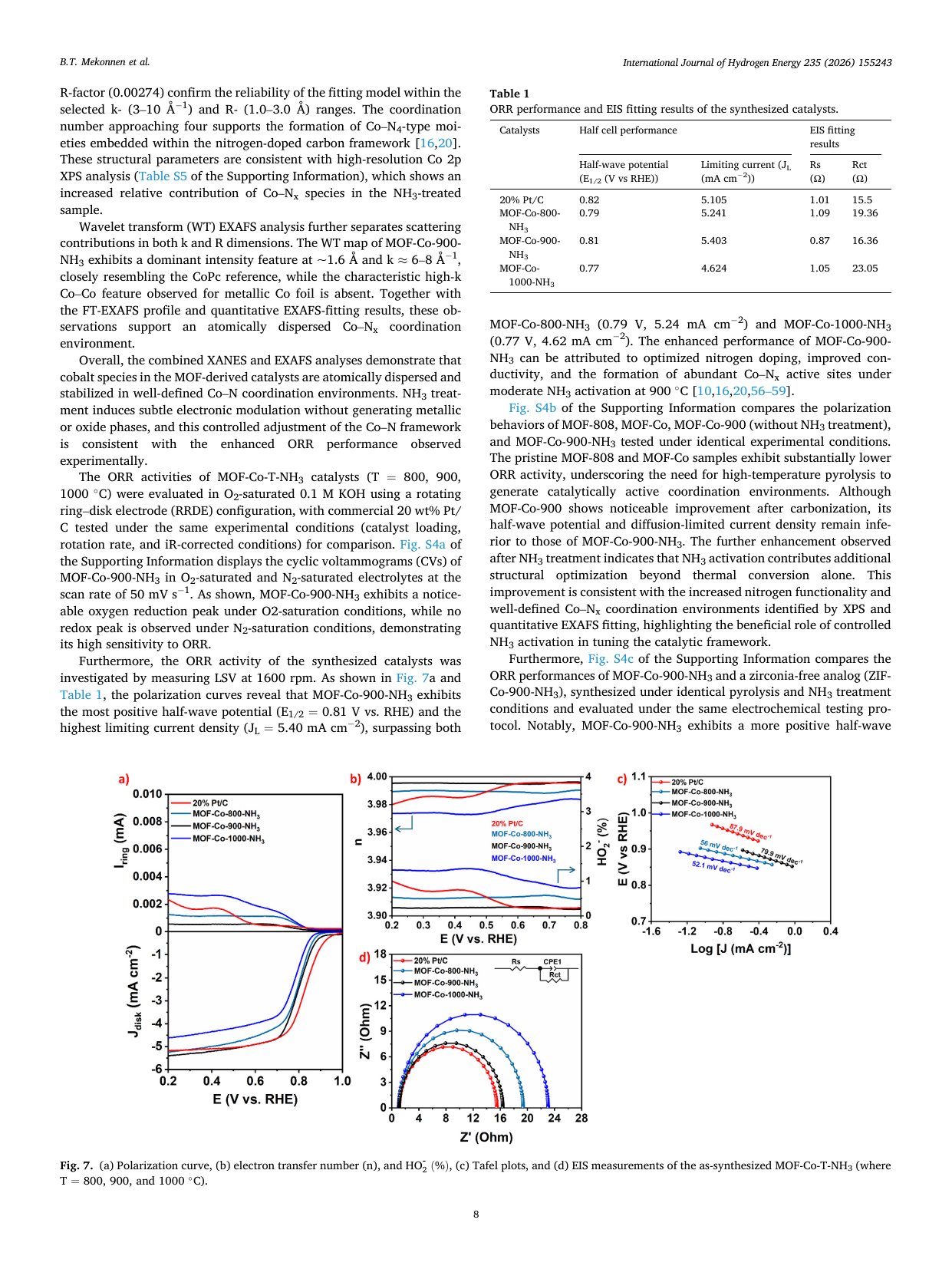
Atomically dispersed Co-Nx sites anchored on t-ZrO2-embedded porous carbon derived from MOF-808 for enhanced oxygen reduction in alkaline media
📄 Abstract
Developing efficient, durable non-precious-metal catalysts for the oxygen reduction reaction (ORR) is critical for alkaline anion-exchange membrane fuel cells (AEMFCs). We report MOF-Co-900-NH3, featuring atomically dispersed Co-Nx sites in a nitrogen-doped carbon matrix anchored on tetragonal ZrO2 nanodomains. Synthesized via MOF-templated stabilization and NH3 activation, it exhibits a half-wave potential of 0.81 V and exceptional durability (only a 35 mV shift after 30,000 cycles), outperforming commercial Pt/C. In AEMFC tests, it delivers a peak power density of 359.2 mW cm-2, surpassing the Pt/C benchmark. Spectroscopic analyses reveal that NH3 activation optimizes the electronic structure of Co-Nx moieties, thereby improving charge transfer. This superior performance stems from the synergy among enriched Co-Nx sites, robust oxide-carbon interfaces, and hierarchical porosity, thereby establishing a viable strategy for next-generation Pt-free electrocatalysts.
🔬 Key Findings
📊 Key Figures