Local collaboration between oxygen vacancy, palladium and platinum trimer triggers exceptional performance in the alkaline fuel cell
氧缺陷、鈀與鉑三聚體的局域協作觸發鹼性燃料電池卓越性能
📄 英文摘要
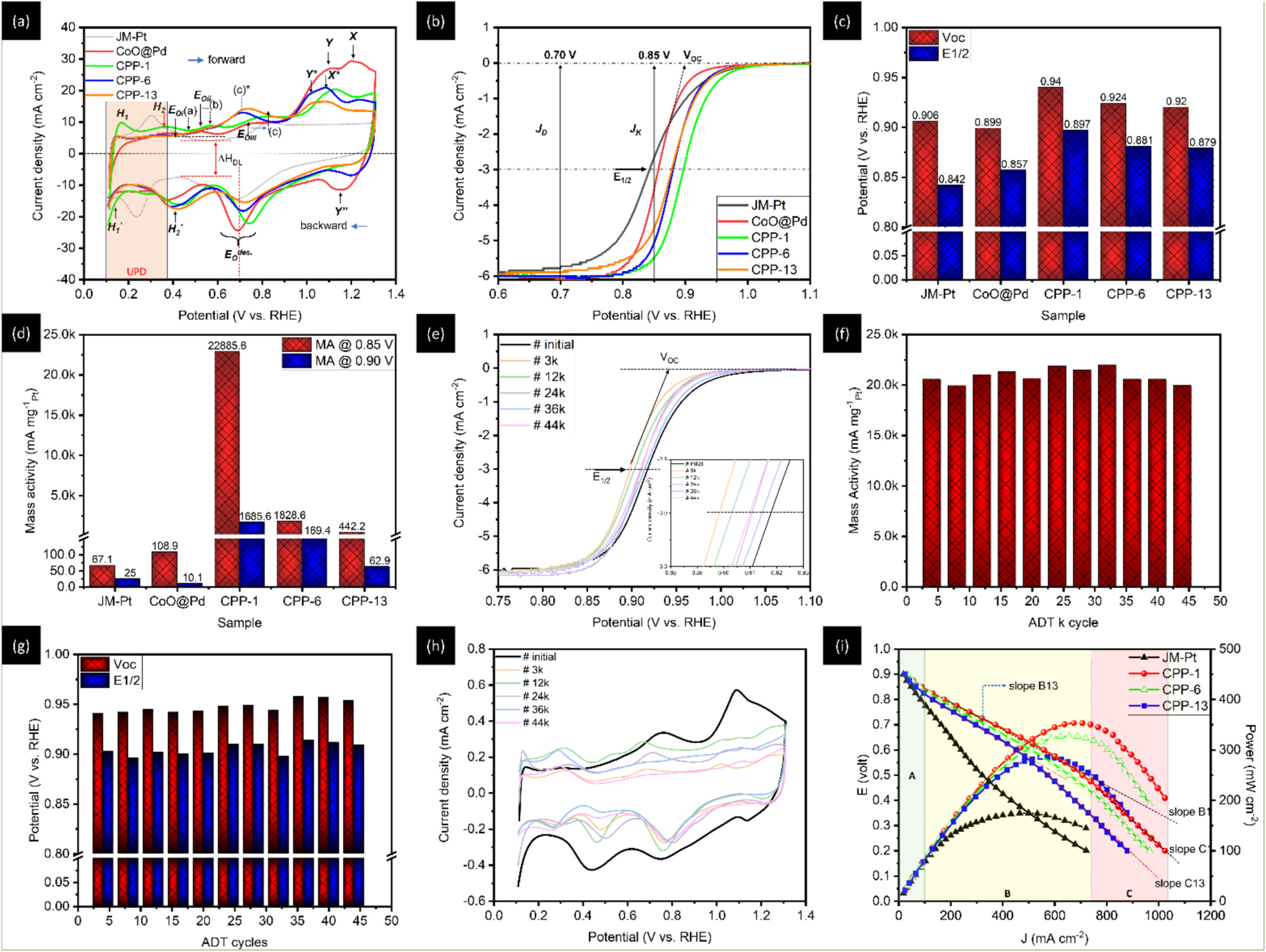
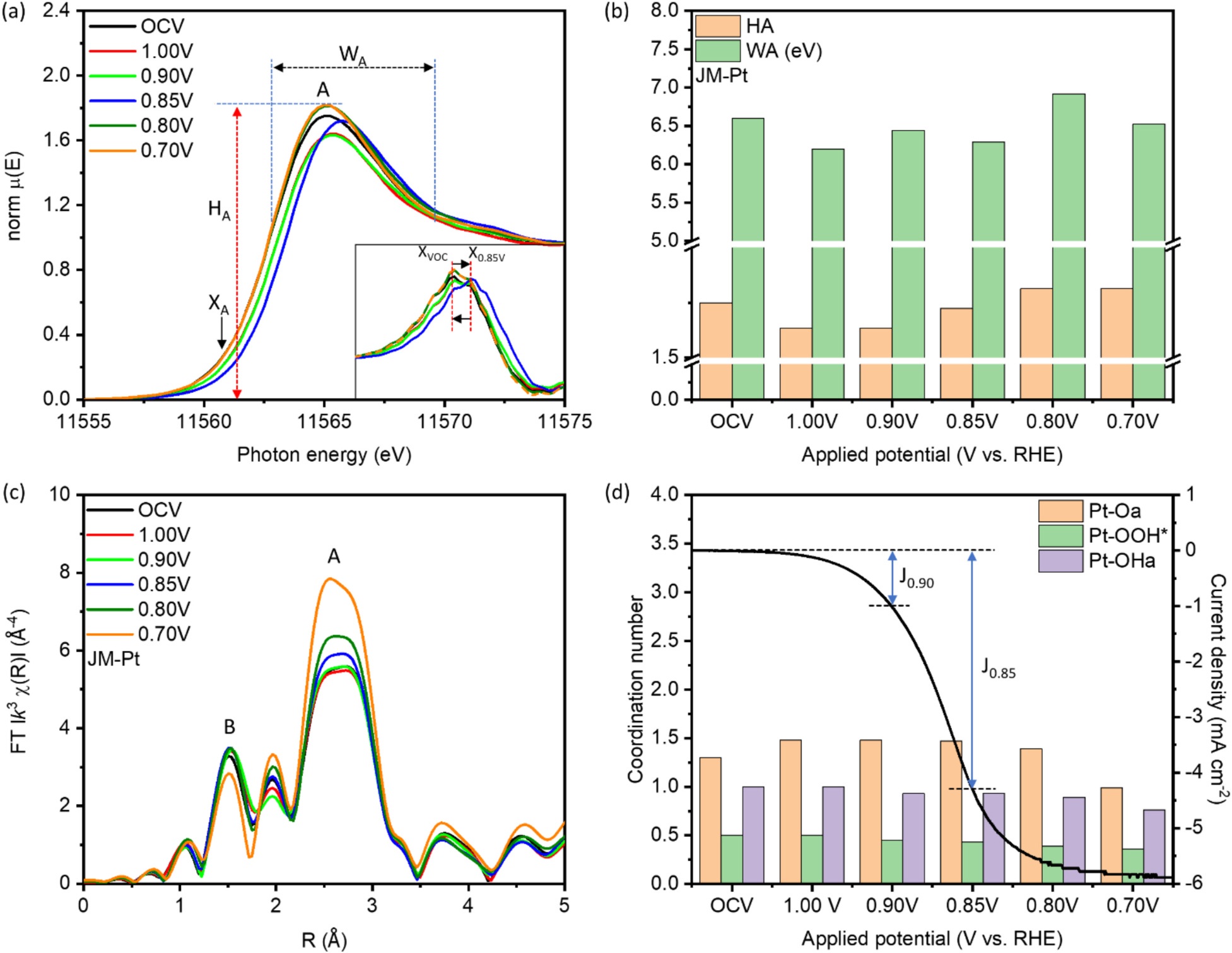
This study highlights a localized synergistic interaction among Pt trimers (PtT), oxygen vacancies (OV), and adjacent Pd atoms within a hierarchically structured catalyst. The catalyst, composed of cobalt oxide-supported palladium nanoparticles (NPs), is specifically optimized for the oxygen reduction reaction (ORR). With a remarkably low platinum loading of approximately 1 wt%, the catalyst (designated as CPP-1) demonstrates exceptional mass activities of 22,885.6 mA mgPt⁻¹ at 0.85 V and 1,685.6 mA mgPt⁻¹ at 0.90 V versus the reversible hydrogen electrode (RHE) in an alkaline (0.1 M KOH) ORR environment. These performance metrics surpass those of commercial platinum catalysts (J.M.-Pt/C; 20 wt% Pt) by factors of 341.6 and 71.7, respectively. Additionally, the PtT enable a maximum power output of 353.2 mW cm⁻² and a current density of 1,024.1 mA cm⁻² in an alkaline fuel cell (AFC) stack. In situ X-ray spectroscopy and electrochemical analysis reveal that PtT facilitates O₂ dissociation into chemisorbed oxygen (Oads), while Pd supports Oads diffusion, and oxygen vacancies (OV) drive the subsequent reduction steps. This coordinated mechanism ensures the simultaneous operation of all intermediate pathways and enables the regeneration of both active PtT sites and OV.
📄 中文摘要
本研究揭示了鉑三聚體(PtT)、氧缺陷(OV)與相鄰鈀原子在一個層級結構觸媒中的局域協同交互作用。該觸媒由氧化鈷負載的鈀奈米粒子(NPs)組成,專為氧還原反應(ORR)優化。在極低的鉑載量(約 1 wt%)下,命名為 CPP-1 的觸媒在鹼性(0.1 M KOH)ORR 環境中,於 0.85 V(相對於可逆氫电极,RHE)展現 22,885.6 mA mgPt⁻¹ 的質量活性,於 0.90 V 展現 1,685.6 mA mgPt⁻¹,分別是商用鉑觸媒(J.M.-Pt/C;20 wt% Pt)的 341.6 倍與 71.7 倍。此外,PtT 使鹼性燃料電池(AFC)堆棧的最大功率輸出達 353.2 mW cm⁻²,電流密度達 1,024.1 mA cm⁻²。原位 X 光譜與電化學分析表明:PtT 促進 O₂ 解離為化學吸附氧(Oads),Pd 協助 Oads 擴散,而 OV 驅動後續的還原步驟。此協調機制確保所有中間途徑同步運行,並使活性 PtT 與 OV 位點得以再生。
🔬 五項核心重要發現
📊 關鍵圖表